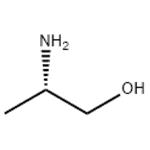
- H-Ala-ol
-
- $0.00/ kg
-
2024-09-23
- CAS:2749-11-3
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1T+
- S-(+)-2-Amino-1-propano
-

- $125.00 / 1ASSAYS
-
2024-08-30
- CAS:2749-11-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 100kg
- L-2-amino-1-propanol
-

- $0.00 / 25Kg/Drum
-
2023-01-31
- CAS:2749-11-3
- Min. Order: 1Kg/Drum
- Purity: 99%
- Supply Ability: 5000KG
|
| | L-Alaninol Basic information |
| | L-Alaninol Chemical Properties |
| Melting point | -2°C | | Boiling point | 72-73 °C11 mm Hg(lit.) | | alpha | 21.8 º (c=2,ethanol) | | density | 0.965 g/mL at 25 °C(lit.) | | refractive index | n20/D 1.450 | | Fp | 145 °F | | storage temp. | Keep in dark place,Inert atmosphere,2-8°C | | Water Solubility | Completely miscible in water | | solubility | Chloroform, Methanol (Slightly) | | form | Oily Liquid | | pka | 12.88±0.10(Predicted) | | Specific Gravity | 0.96 | | color | Clear colorless to yellow | | optical activity | [α]20/D +18°, neat | | Sensitive | Hygroscopic | | BRN | 1718865 | | CAS DataBase Reference | 2749-11-3(CAS DataBase Reference) | | NIST Chemistry Reference | 1-Propanol, 2-amino-, (S)-(2749-11-3) |
| | L-Alaninol Usage And Synthesis |
| Chemical Properties | Colorless liquid | | Uses |
(S)-(+)-2-Amino-1-propanol (L-Alaninol) may be used in the preparation of unsymmetrical tridentate Schiff base ligands via condensation with carbonyl compounds. It may also be used as a chiral auxillary for the preparation of tert-butyl 4-N-[(2-hydroxy-1-(S)-methyl)ethylamino]-2-methylene-4-(S)-phenyl-butyrate.
| | Uses | S-(+)-2-Amino-1-propanol is an aliphatic amino alcohol shown to induce an antiproliferative effect in B16 melanoma cells. S-(+)-2-Amino-1-propanol is used in the preparation of oxazolines which are of
ten used as ligands in homogeneous catalysis. | | Definition | ChEBI: An amino alcohol that is L-alanine in which the carboxy group has been reduced to the corresponding alcohol. | | Biochem/physiol Actions | Pseudomonas sp. strain KIE171 was able to grow with L-alaninol [S-(+)-2-amino-1-propanol] as the sole carbon source[1]. W?sch et al. found that mutant KIE171-BI transformed isopropylamine to L-alaninol, mutant KIE171-BII failed to do so. | | Purification Methods | Purify it as for S-2-amino-3-methylbutan-1-ol below. [Beilstein 4 IV 1615.] | | References | [1] Susana I de Azevedo W?sch. “Transformation of isopropylamine to L-alaninol by Pseudomonas sp. strain KIE171 involves N-glutamylated intermediates.”Applied and Environmental Microbiology (2002): 68.
|
| | L-Alaninol Preparation Products And Raw materials |
|