Uses of Acrylonitrile
Oct 25,2021
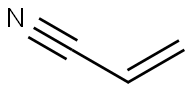
Acrylonitrile is an organic compound with the formula CH2CHCN. It is a colorless volatile liquid although commercial samples can be yellow due to impurities. It has a pungent odor of garlic or onions.In terms of its molecular structure, it consists of a vinyl group linked to a nitrile. It is an important monomer for the manufacture of useful plastics such as polyacrylonitrile. It is reactive and toxic at low doses.

Uses
Acrylonitrile is used in the manufacture of acrylic fibers, plastic surface coatings, adhesives, and synthetic rubbers. It was formerly used as a pesticide fumigant for stored grains. It is a chemical intermediate in the synthesis of antioxidants, pharmaceutical dyes, surface-active agents, and in reactions requiring the cyanoethyl group.
Mechanism of Toxicity
Acrylonitrile is both readily volatile in air and highly soluble in
water. These characteristics determine the behavior of acrylonitrile
in the environment. The principal pathway leading to
the degradation of acrylonitrile in air is photooxidation,
mainly by reaction with hydroxyl radicals (OH).
Acrylonitrile may also be oxidized by other atmospheric components such as ozone and oxygen. Very little is known about the nonbiologically mediated transformation of acrylonitrile in water. It is oxidized by strong oxidants such as chlorine used to disinfect water. Acrylonitrile is readily degraded by aerobic microorganisms in water.
Environmental Fate
Acrylonitrile owes some of its toxicity to cyanide generation,
which inhibits cellular respiration. Preinduction of microsomal
mixed function oxidase (MFO) with Aroclor 1254 greatly
enhanced the toxicity of acrylonitrile and caused a threefold
increase in cyanide levels in rats.
Therefore, metabolic activation appears to be necessary in the toxicity of acrylonitrile. The direct reaction of acrylonitrile with the SH groups of proteins and its epoxide metabolite are also expected to be responsible for its effects. Acrylonitrilehas also been associated with oxidative stress.
- Related articles
- Related Qustion
- Acrylonitrile - a Brief Introduction Apr 27, 2022
Acrylonitrile, also known as AN or vinyl cyanide, is a man-made VOC. It is a pungent smelling, colorless flammable liquid.
- Acrylonitrile-Health Hazard and Toxicity Sep 4, 2019
Acrylonitrile is an organic compound with the formula CH2CHCN. It is a colorless volatile liquid, although commercial samples can be yellow due to impurities. In terms of its molecular structure, it consists of a vinyl group linked to a nit
Acrylic acid, also known as prop-2-enoic acid, is an organic molecule with the formula C3H4O2, and is the simplest of the unsaturated carboxylic acids. At room temperature, acrylic acid is a colorless liquid with a tart or acrid smell. Indu....
Oct 22,2021Organic ChemistryEthylene oxide, also known as oxane, ethylene oxide, and ETO, it is a strong alkylating agent that can readily react with cellular nucleophiles to inactivate a variety of cellular macromolecules.....
Oct 25,2021Organic reagentsAcrylonitrile
107-13-1You may like
- Acrylonitrile
-

- $1.00 / 25KG
- 2025-10-21
- CAS:107-13-1
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 10 mt
- Acrylonitrile
-

- $0.00 / 25KG
- 2025-08-08
- CAS:107-13-1
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 50000KG/month
- Acrylonitrile
-

- $10.50 / 1KG
- 2025-05-26
- CAS:107-13-1
- Min. Order: 1KG
- Purity: 99.9%
- Supply Ability: 10 ton