| Identification | More | [Name]
Tetrahydrofurfuryl bromide | [CAS]
1192-30-9 | [Synonyms]
2-(BROMOMETHYL)TETRAHYDROFURAN
TETRAHYDROFURFURYL BROMIDE
TIMTEC-BB SBB003909
2-(bromomethyl)tetrahydro-fura
tetrahydro-2-(bromomethyl)-fura
TETRAHYDROFRUFRYL BROMIDE
Tetrahydrofurfuryl bromide, pract., 95%
Tetrahydrofurfuryl bromide, tech., 90%
tetrahydro-2-(bromomethyl)-furan
A-(BROMOMETHYL)TETRA HYDROFURAN
(RS)-Tetrahydrofurfuryl bromide
2-(Bromomethyl)oxolane
Tetrahydrofurfuryl bromide, 90%, tech. | [EINECS(EC#)]
214-750-3 | [Molecular Formula]
C5H9BrO | [MDL Number]
MFCD00016894 | [Molecular Weight]
165.03 | [MOL File]
1192-30-9.mol |
| Chemical Properties | Back Directory | [Appearance]
Colorless to light yellow liqui | [Boiling point ]
170-171 °C (755 mmHg) | [density ]
1.45
| [refractive index ]
1.4853-1.4873
| [Fp ]
63 °C
| [storage temp. ]
Refrigerator | [solubility ]
Chloroform (Slightly), Methanol (Slightly) | [form ]
Oil | [color ]
Clear Light Yellow | [BRN ]
102717 | [InChI]
InChI=1S/C5H9BrO/c6-4-5-2-1-3-7-5/h5H,1-4H2 | [InChIKey]
VOHILFSOWRNVJJ-UHFFFAOYSA-N | [SMILES]
O1CCCC1CBr | [CAS DataBase Reference]
1192-30-9(CAS DataBase Reference) | [EPA Substance Registry System]
1192-30-9(EPA Substance) |
| Safety Data | Back Directory | [Hazard Codes ]
Xi | [Risk Statements ]
R36/37/38:Irritating to eyes, respiratory system and skin . | [Safety Statements ]
S24/25:Avoid contact with skin and eyes . | [RIDADR ]
1993 | [RTECS ]
LU6000000 | [TSCA ]
Yes | [HS Code ]
29321900 | [Toxicity]
mouse,LD50,intravenous,180mg/kg (180mg/kg),U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#03416, |
| Hazard Information | Back Directory | [Chemical Properties]
Colorless to light yellow liqui | [Uses]
(±)-Tetrahydrofurfuryl Bromide is a general chemical reagent used in the synthesis of tubulin polymerization inhibitors. | [Synthesis]
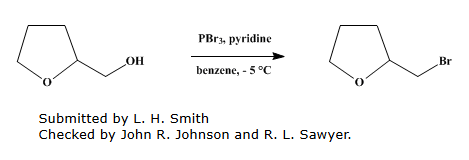
In a 500-ml three-necked flask equipped with a stirrer, thermometer, dropping funnel, and calcium chloride tube, place 96 g of redistilled PBr₃ and 50 ml of dry benzene. Add 15 g of dry pyridine dropwise with stirring over 15 minutes.
Cool the flask in an ice-salt bath to -5°C. Then, slowly add a mixture of 102 g of tetrahydrofurfuryl alcohol and 5 g of dry pyridine over 4 hours, maintaining the internal temperature between -5° and -3°C. Stir for an additional hour and allow the mixture to warm to room temperature.
Let the mixture stand for 24-48 hours. Transfer it to a Claisen flask, rinse with a little benzene, and remove the benzene by distillation under reduced pressure (bath temperature ≤90°C). Finally, distill the product under high vacuum (5-10 mm Hg) with the bath temperature up to 150-155°C.
The crude product is redistilled to give pure tetrahydrofurfuryl bromide, boiling at 69-70°C/22 mm Hg. The yield is 90-102 g (53-61%)[1].
 | [References]
[1] Org. Synth. 1943, 23, 88. DOI: 10.15227/orgsyn.023.0088. |
|
|