| Identification | More | [Name]
Bupivacaine | [CAS]
2180-92-9 | [Synonyms]
BUPIVACAINE
BUPIVACAINE BASE
MARCAINE
(.+/-.)-1-Butyl-2',6'-pipecoloxylidide
1-Butyl-2-(2,6-xylycarbamoyl)piperidine
1-Butyl-2',6'-pipecoloxylidide
1-butyl-2’,6’-pipecoloxylidide
1-butyl-n-(2,6-dimethylphenyl)-2-piperidinecarboxamid
1-Butyl-N-(2,6-dimethylphenyl)-2-piperidinecarboxamide
2',6'-Pipecoloxylidide, 1-butyl-
6’-pipecoloxylidide,1-butyl-2
Anekain
Carbostesin
DL-Bupivacaine
LAC-43
WIN 11318
win11318 | [EINECS(EC#)]
218-553-3 | [Molecular Formula]
C18H28N2O | [MDL Number]
MFCD00243007 | [Molecular Weight]
288.43 | [MOL File]
2180-92-9.mol |
| Hazard Information | Back Directory | [Originator]
Carbostesin,Astra,W. Germany,1967 | [Uses]
Anesthetic (local). | [Uses]
Like lidocaine and mepivacaine, bupivacaine is used in infiltration, spinal, and epidural anes�thesia in blocking nerve transmission. Its most distinctive property is its long-lasting action.
It is used for surgical intervention in urology and in lower thoracic surgery from 3 to 5 h in
length, and in abdominal surgery lasting from 45 to 60 min. It is used to block the trifacial
nerve, the sacral and brachial plexuses, in resetting dislocations, in epidural anesthesia, and
during Cesarian sections. | [Definition]
ChEBI: 1-butyl-N-(2,6-dimethylphenyl)piperidine-2-carboxamide is a piperidinecarboxamide obtained by formal condensation of the carboxy group of N-butylpipecolic acid with the amino group of 2,6-dimethylaniline. It is a piperidinecarboxamide, an aromatic amide and a tertiary amino compound. It is a conjugate base of a 1-butyl-2-[(2,6-dimethylphenyl)carbamoyl]piperidinium. | [Manufacturing Process]
121 parts by weight of 2.6-xylidine are heated with 400 parts of
diethylmalonate at 160°C for 1 hour, and the alcohol formed by the reaction is
allowed to distill off. Thereafter the reaction mass is cooled to 80°C, and 500
parts of alcohol are added. After cooling the dixylidide is sucked off, and the
alcohol solution with malonic ester monoxylidide is poured into 2,000 parts of
water. The monoxylidide precipitates, is filtered off and washed with water,
and recrystallized in diluted alcohol. Nitrosation thereafter takes place by
dissolving the dried monoxylidide in chloroform and by introducing nitrosyl
chloride at 0°C until the nitrosation is completed. The isonitrosomalonic ester
xylidide is filtered off and dried. Thereafter the reduction takes place with zinc
powder and formic acid at 90°-100°C.
The formic acid is distilled off, and the remainder dissolved in warm benzene
and washed with a bicarbonate solution to a neutral reaction. After the
benzene has been distilled off, the aminomalonic ester xylidide is obtained.
This is treated with an equal quantity of sodium ethylate and boiled with twice
the theoretical quantity of tetramethylene bromide in absolute alcohol.
After 6 hours of boiling, the sodium bromide formed is separated, and the
mixture is steamdistilled in order to remove the excess of tetramethylene
bromide. The remaining oil, which mainly consists of deltabromobutylaminomalonic
ester xylidide is separated from the water and boiled
with 3 parts of concentrated hydrochloric acid for 3 hours. Thereafter carbonfiltering
and evaporation to dryness under vacuum takes place. The residue is
dissolved in water, and the pH adjusted with sodium hydroxide to 5.5. The
solution is extracted twice with ether, and the water is made strongly alkaline
with sodium hydroxide.
The oil precipitates and is crystallized after a time. The crystals are separated
and dried under vacuum. The pipecolyl-2,6-xylidide produced is alkylated by
boiling for 10-20 hours with 0.6 part n-butylbromide in an n-butanol solution
in the presence of 0.5 part potassium carbonate. The potassium carbonate is
filtered off and the butanol is distilled off in vacuum. The residue is dissolved
in diluted hydrochloric acid and carbon treated, after which the base is
precipitated with sodium hydroxide in the form of white crystals, which are
filtered off and washed with water. The base obtained, which consists of N-n-butyl-pipecolyl-2,6-xylidide is sufficiently pure for the production of salts. | [Brand name]
Marcaine (Hospira); Sensorcaine
(AstraZeneca). | [Therapeutic Function]
Local anesthetic | [General Description]
Bupivacaine was synthesized simultaneously with mepivacainein 1957 but was at first overlooked because of the increasedtoxicity compared with mepivacaine. When themethyl on the cyclic amine of mepivacaine is exchanged fora butyl group the lipophilicity, potency and the duration ofaction all increase. Literature reports of cardiovascular toxicity,including severe hypotension and bradycardia, areabundant in the literature.91 Bupivacaine is highly bound toplasma proteins (95%), and thus the free concentration mayremain low until all of the protein binding sites are occupied.After that point, the plasma levels of bupivacaine rise rapidlyand patients may progress to overt cardiac toxicity withoutever showing signs of CNS toxicity. The cardiotoxicity ofbupivacaine is a result of its affinity to cardiac tissues and itsability to depress electrical conduction and predispose theheart to reentry types of arrhythmias. The cardiotoxicity ofbupivacaine was found to be significantly more prominentwith the “R” isomer, or the racemic mixture, thus the “S”stereoisomer is now on the market as levobupivacaine. | [Mechanism of action]
Bupivacaine is a local anaesthetic containing a chiral centre and adopts dextro and laevo forms. The enantiopure l form is less cardio- and neurotoxic and has an equivalent potency to the racemic mixture; therefore levobupivacaine is often preferred to reduce the potential for toxicity. Stereoselectivity describes the differences in response at a given receptor for the different enantiomers (such as the response discussed for S(+) ketamine). The opioid and NMDA receptors also exhibit stereoselectivity. | [Pharmacology]
Bupivacaine is a chiral compound used clinically for 50 years, with a slower
onset, greater potency and longer duration of action than lidocaine. Initial
benefits of bupivacaine were sensory–motor separation and minimal
tachyphylaxis, unlike repeated doses of lidocaine. However, it has greater
potential for cardiac toxicity, related to its avid binding to and slow
dissociation from cardiac N a+ channels. Inadvertent intravenous
administration may result in systemic toxicity (see later), and it is
contraindicated for intravenous regional anaesthesia.
Bupivacaine is commonly used for epidural administration in obstetrics and
postoperative pain management. A hyperbaric preparation containing
80 mg ml-1 glucose is available for spinal anaesthesia. | [Side effects]
Common side effects of bupivacaine include:
weakness, long-lasting numbness or tingling;
feeling restless or drowsy;
tremors;
headache, blurred vision;
fast or slow heartbeats;
breathing problems;
chills or shivering;
back pain;
nausea, vomiting. | [Synthesis]
Bupivacaine, N-2,6-(dimethyl)1-butyl-2-piperidincarboxamide (2.2.7), is chemically similar to mepivacaine and only differs in the replacement of the N-methyl sub�stituent on the piperidine ring with an N-butyl substituent. There are also two suggested meth�ods of synthesis. The first comes from α-picolin-2,6-xylidide (2.2.4). The alkylation of the last with butyl bromide gives the corresponding pyridine salt (2.2.6). Finally, it is reduced by hydrogen using platinum oxide as a catalyst into a piperidine derivative—bupivacaine.
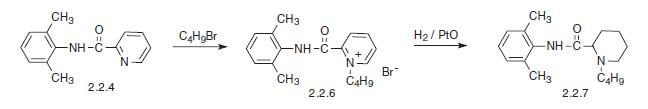
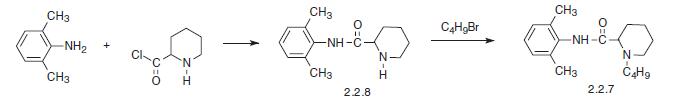
The other method results directly from the piperidine-2-carboxylic acid chloride, which is reacted with 2,6-dimethylaniline. The resulting amide (2.2.8) is further alkylated with butyl bromide to bupivacaine [17–19].
|
|
|