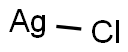Silver chloride
- CAS No.
- 7783-90-6
- Chemical Name:
- Silver chloride
- Synonyms
- AgCl;ver(I) ChL;Chlorosilver;Silver chlorid;SILVER CHLORIDE;Silver(Ⅰ)Chloride;silvermonochloride;SILVER (I) CHLORIDE;Silver monochloride;Silver chloride 99+
- CBNumber:
- CB3151552
- Molecular Formula:
- AgCl
Lewis structure

- Molecular Weight:
- 143.32
- MDL Number:
- MFCD00003399
- MOL File:
- 7783-90-6.mol
- MSDS File:
- SDS
| Melting point | 455 °C (lit.) |
|---|---|
| Boiling point | 1550 °C |
| Density | 5.56 |
| vapor pressure | 1 mm Hg ( 912 °C) |
| refractive index | 2.071 |
| Flash point | 1550°C |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 0.00188g/l |
| form | beads |
| color | Yellow |
| Specific Gravity | 5.56 |
| Water Solubility | 1.93 mg/L (25 ºC) |
| Sensitive | Light Sensitive |
| Merck | 14,8509 |
| Solubility Product Constant (Ksp) | pKsp: 9.75 |
| Dielectric constant | 11.2(0.0℃) |
| Stability | Stable, but discolours in light. |
| InChIKey | HKZLPVFGJNLROG-UHFFFAOYSA-M |
| CAS DataBase Reference | 7783-90-6(CAS DataBase Reference) |
| Indirect Additives used in Food Contact Substances | SILVER CHLORIDE |
| EWG's Food Scores | 2 |
| FDA UNII | MWB0804EO7 |
| NIST Chemistry Reference | Silver chloride(7783-90-6) |
| EPA Substance Registry System | Silver chloride (7783-90-6) |
| Pesticides Freedom of Information Act (FOIA) | Silver chloride |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H290-H410 | |||||||||
| Precautionary statements | P234-P273-P390-P391-P501 | |||||||||
| Hazard Codes | N | |||||||||
| Risk Statements | 50/53-50 | |||||||||
| Safety Statements | 24/25-61-60 | |||||||||
| RIDADR | UN 3077 9 / PGIII | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | VW3563000 | |||||||||
| F | 8 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 2843 29 00 | |||||||||
| HazardClass | 9 | |||||||||
| Toxicity | LD50 orally in Rabbit: > 5110 mg/kg | |||||||||
| NFPA 704 |
|
Silver chloride price More Price(47)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 204382 | Silver chloride 99.999% trace metals basis | 7783-90-6 | 1g | $33.6 | 2024-03-01 | Buy |
| Sigma-Aldrich | 204382 | Silver chloride 99.999% trace metals basis | 7783-90-6 | 5g | $79.9 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1.19203 | Silver chloride 99+ for analysis 99+ EMSURE? | 7783-90-6 | 25g | $719 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1.19203 | Silver chloride 99+ for analysis 99+ EMSURE? | 7783-90-6 | 100g | $2160 | 2024-03-01 | Buy |
| Alfa Aesar | 010857 | Silver chloride, Premion?, 99.997% (metals basis) | 7783-90-6 | 25g | $206 | 2024-03-01 | Buy |
Silver chloride Chemical Properties,Uses,Production
Physical Properties
White granular powder or cubic crystals; refractive index 2.071; darkens on exposure to light; density 5.56 g/cm3; Moh’s hardness 2.5; melts at 455°C; vaporizes at 1,547°C; vapor pressure 1 and 5 torr at 912 and 1,019°C; insoluble in water, alcohol and dilute acids; soluble in ammonia solution and concentrated sulfuric acid, alkali cyanide, ammonium carbonate; also soluble in potassium bromide and sodium thiosulfate solutions.
Crystal System
The space lattice of AgCl belongs to the cubic system, and its rock salt structure has a lattice constant of a=0.554 nm, Ag–Cl=0.277 nm. Cleavage does not occur.
Uses
Silver chloride is used in silver plating and to obtain pure silver. The salt also finds applications in photography and optics; in photochromic glass; and in electrodes and batteries. It is used to make antiseptic silver solution. It occurs as the mineral cerargyrite.
Preparation
Silver chloride is prepared by slowly adding an alkali metal chloride solution to a hot solution of silver nitrate. The solution mixture is boiled:
Ag+ (aq) + Cl¯ (aq) → AgCl (s)
The precipitate is washed with hot water. The product is purified by dissolving in ammonia solution, filtering out any insoluble residues, and then adding hydrochloric acid to reprecipitate silver chloride. Preparation should be carried out in the dark in ruby red light.
Chemical Properties
Silver chloride, AgCl, is a white,granular powder that darkens on exposure to light,finally turning black.It exists in several modifications differing in behavior toward light and solubility in various solvents. Soluble in ammonium hydroxide, concentrated sulfuric acid, and sodium thiosulfate and potassium bromide solutions, very slightly soluble in water, can be melted, cast, and fabricated like a metal. Derived by heating a silver nitrate solution and adding hydrochloric acid or salt solution. The whole is boiled, then filtered. This must take place in the dark or under a ruby-red light. Used in photography,photometry and optics, batteries, photochromic glass,silver plating,production of pure silver, and as an antiseptic. Single crystals are used for infrared absorption cells and lens elements and as a lab reagent
Uses
In silver plating, in making antiseptic silver preparations.
Uses
Used in photographic films, to coat and silver glass, as an antiseptic, and to absorb infrared light in lenses.
Uses
Found in nature as horn silver, this white powder is made by the combination of a soluble chloride and silver nitrate. Silver bromide could also be formed by exposing metallic silver to the fumes of bromine as in the daguerreotype process. It is soluble in sodium thiosulfate, potassium bromide solutions, and strong ammonia. This silver halide was the first to be observed to darken spontaneously by exposure to light. Silver chloride formed the basis of the photogenic drawing, salted paper print, albumen print, collodion-chloride POP, gelatin chloride POP, and gaslight paper.
Uses
Employed in Silver plating. Owing to its characteristic of reversible reduction to silver metal, it is used in photochromic lenses. Used as a cathode in sea water activated batteries. In electrochemistry, silver chloride electrode is used for potentiometric measurements. It serves as an antidote for mercury poisoning, and eliminates mercury from body. It is used in glass manufacturing industry. It is useful in the production of bandages, wound healing products and inglaze lustre, personal deodorant products, as well as for long term preservation of drinking water in water tanks; its pharmaceutical composition finds use as an antibacterial agent.
Application
?AgCl is very important as a linear polarizer in the infrared region (λ: 2–23 mm). The refractive index is almost constant in the infrared region and the polarization angle is almost independent of wavelength. The polarization angles are 63°43' (3 mm), 63°20' (10 mm), and 63°33' (20 mm), showing the difference of angle below 18 for λ: 2–23 mm. The polariscope is fabricated typically by arranging the six sheets of plates with the thickness of 0.5 mm in the shape of roof type. Bakelite or plastic is good for the material of the holder case.
Definition
T3DB: Silver chloride is a chloride of silver that occurs naturally as the mineral chlorargyrite. It is used to make photographic paper and pottery glazes. It is also found in stained glass colorants, bandages, and other wound healing products, and may be used as an antidote to mercury poisoning. Silver is a metallic element with the chemical symbol Ag and atomic number 47. It occurs naturally in its pure, free form, as an alloy with gold and other metals, and in minerals such as argentite and chlorargyrite.
Hazard
As for silver.
Health effects
Silver itself is not toxic to humans, but most silver salts are. In large doses, silver and compounds containing it can be absorbed into the circulatory system and become deposited in various body tissues, leading to argyria, which results in a blue-grayish pigmentation of the skin, eyes, and mucous membranes. Argyria is rare, and although, so far as known, this condition does not otherwise harm a person's health, it is disfiguring and usually permanent. Mild forms of argyria are sometimes mistaken for cyanosis. Exposure to high levels of silver in the air has resulted in breathing problems, lung and throat irritation, and stomach pains. Skin contact with silver can cause mild allergic reactions such as rash, swelling, and inflammation in some people.
Flammability and Explosibility
Non flammable
Toxicology
Metallic silver is oxidized and may deposit in the tissues, causing arygria. The silver ion is known to inhibit glutathione peroxidase and NA+,K+-ATPase activity, disrupting selenium-catalyzed sulfhydryl oxidation-reduction reactions and intracellular ion concentrations, respectively. Silver nanoparticles are believed to disrupt the mitochondrial respiratory chain, causing oxidative stress, reduced ATP synthesis, and DNA damage.
Purification Methods
Recrystallise it from conc NH3 solution by acidifying with HCl, filtering off the solid, washing it with H2O and drying it in a vacuum. It is soluble in NH3 and should be kept in the dark.
Silver chloride Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-13102810335 | admin@hbsaisier.cn | China | 892 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 | sales@hbmojin.com | China | 12456 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 | peter@yan-xi.com | China | 5993 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 | hbjbtech@163.com | China | 1000 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 16803 | 58 |
| Shanghai Bojing Chemical Co.,Ltd. | +86-86-02137122233 +8613795318958 | bj1@bj-chem.com | China | 298 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21689 | 55 |
| career henan chemical co | +86-0371-86658258 | sales@coreychem.com | China | 29914 | 58 |
| SHANDONG ZHI SHANG CHEMICAL CO.LTD | +86 18953170293 | sales@sdzschem.com | China | 2931 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
Related articles
- Is silver chloride soluble in water? is it used for?
- Silver chloride is a white crystalline solid that is well known for its low solubility in water and its sensitivity to light.
- Mar 7,2024
View Lastest Price from Silver chloride manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-05-06 | Silver chloride
7783-90-6
|
US $15.00-2.00 / kg | 1kg | 99% | 300tons | Hebei Dangtong Import and export Co LTD | |
 |
2024-04-12 | Silver chloride
7783-90-6
|
US $6.00 / KG | 1KG | More than 99% | 500KG/Month | Hebei Saisier Technology Co., LTD | |
 |
2024-03-27 | Silver chloride
7783-90-6
|
US $5.00-0.10 / KG | 1KG | 98% | g-kg-tons, free sample is available | Henan Fengda Chemical Co., Ltd |
-

- Silver chloride
7783-90-6
- US $15.00-2.00 / kg
- 99%
- Hebei Dangtong Import and export Co LTD
-

- Silver chloride
7783-90-6
- US $6.00 / KG
- More than 99%
- Hebei Saisier Technology Co., LTD
-
- Silver chloride
7783-90-6
- US $5.00-0.10 / KG
- 98%
- Henan Fengda Chemical Co., Ltd