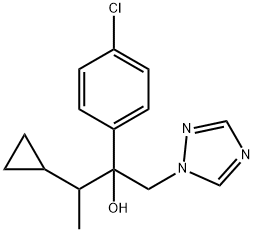
Cyproconazole
- CAS No.
- 94361-06-5
- Chemical Name:
- Cyproconazole
- Synonyms
- cyproconazole (ISO) (2RS,3RS2RS,3SR)-2-(4-chlorophenyl)-3-cyclopropyl-1-(1H-1,2,4-triazol-1-yl)butan-2-ol;ALTO;CS-142;SHANDON;Alto 100;Alto 100SL;CYPROCONAZOLE;Enavogliflozin Impurity 40;(113096-99-4) cyproconazole;Cyproconazole @100 μg/mL in MeOH
- CBNumber:
- CB3716690
- Molecular Formula:
- C15H18ClN3O
- Molecular Weight:
- 291.78
- MDL Number:
- MFCD01678672
- MOL File:
- 94361-06-5.mol
- MSDS File:
- SDS
| Melting point | 108℃ |
|---|---|
| Boiling point | >250℃ |
| Density | 1.32 |
| vapor pressure | 3.46 x l0-5 Pa (20 °C) |
| refractive index | 1.6330 (estimate) |
| Flash point | >100 °C |
| storage temp. | Store at -20°C |
| solubility | Soluble in acetone, ethanol, xylene, and dimethyl sulfoxide. |
| Water Solubility | 140 mg l-1(25 °C) |
| pka | 12.59±0.29(Predicted) |
| form | Solid |
| color | White to off-white |
| BRN | 8396421 |
| Stability | Hygroscopic |
| LogP | 3.1 at 25℃ and pH7.2 |
| CAS DataBase Reference | 94361-06-5(CAS DataBase Reference) |
| EWG's Food Scores | 3 |
| FDA UNII | 622B9C3E6T |
| Pesticides Freedom of Information Act (FOIA) | Cyproconazole |
| EPA Substance Registry System | Cyproconazole (94361-06-5) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS06,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301-H360D-H373-H410 | |||||||||
| Precautionary statements | P201-P202-P260-P264-P273-P301+P310 | |||||||||
| Hazard Codes | Xn,N | |||||||||
| Risk Statements | 22-50/53-63 | |||||||||
| Safety Statements | 36/37-60-61-36 | |||||||||
| RIDADR | UN3077 9/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | XZ4803250 | |||||||||
| HazardClass | 9 | |||||||||
| Toxicity | LD50 in male and female rats (mg/kg): 1020, 1330 orally; in rats: 2000 dermally; LD50 in carp: 18.9 mg/l; in trout 7.2 mg/l; in bluegill sunfish 6.0 mg/l in water; LD50 in bobwhite quail (mg/kg): 150 orally; LD50 (8 day dietary) in bobwhite quail, mallard duck: 816, 1197 mg/kg (Gisi, 1986) | |||||||||
| NFPA 704 |
|
Cyproconazole price More Price(17)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 46068 | Cyproconazol mixture of diastereomers | 94361-06-5 | 100mg | $147 | 2024-03-01 | Buy |
| Alfa Aesar | J63142 | Cyproconazole | 94361-06-5 | 5g | $109.65 | 2024-03-01 | Buy |
| Alfa Aesar | J63142 | Cyproconazole | 94361-06-5 | 10g | $153.65 | 2024-03-01 | Buy |
| TRC | C989070 | Cyproconazole | 94361-06-5 | 250mg | $135 | 2021-12-16 | Buy |
| Usbiological | 163261 | Cyproconazole | 94361-06-5 | 250mg | $403 | 2021-12-16 | Buy |
Cyproconazole Chemical Properties,Uses,Production
Physical and chemical properties
It appears as colorless crystals with the melting point of 106 to 109 °C, the boiling point of> 250 °C and the vapor pressure at 20 ° C of 0.03470 mPa. It has a relative density of 1.259 with water solubility at 25 ℃ of 1.4g/kg, acetone> 2300g/kg, dimethyl sulfoxide> 180g/kg, ethanol> 230g/kg and xylene: 1200g/kg. It has a Ko819 (±195) (pH 7). Under 70 °C, it can be stable up to 15 days. The DT50 of the soil surface was 21 days under the sunlight; pH value is at the range of 3~9. It is stable under 59 ℃. The soil adsorption coefficient K was 4.1 (loam, pH 6.4, organic matter content: 2.3%), 16 (soil sandy soil, pH 5.1, organic matter content: 3.9%).
Fungicide
Cyproconazole is a kind of triazole agricultural fungicide with the mechanism of action being inhibition of sterol demethylation with prevention and treatment effect. Currently it has been widely used in pesticides, being effective in the treatment of powdery mildew, rust fungi, Rhizopus, Cercospora, Acipenseris and Scleroderma in cereal crops, coffee, sugar beet and fruit trees. It is effective in the treatment of following plant diseases such as the rust diseases of cereals and coffee, powdery mildew diseases of cereals, fruit trees and grape, leaf spot of peanuts and beet, apple scab and white rot disease cereal eye spot, leaf spot and reticulosis of peanuts.
Its persistence in the treatment of wheat rust disease is about 4 to 6 weeks and of powdery mildew is 3 to 4 weeks. The spraying method is as follows: spray, the dosage is 80g/ha for cereal crops, 20-50g/ha for coffee, 40-60g/ha for beets and 10 g/ha for fruit tree. For example, we can apply 40~100g/ha for prevention of cereals and coffee rust, cereal, fruit powdery mildew and so on. Cyproconazole can be mixed with other fungicides for better control of cereal eye disease, leaf spot and leaf net blotch disease.
Analytical method: The product was analyzed by GLC or HPLC. The residue was assayed by GLC.
Allowable residue: The residual amount of the parent compound in the cereal crop is 0.03 mg/kg.
Toxicity
Male rats had acute oral LD50 of 1020 mg/kg, female rats of 1333 mg/kg; male mice of 200 mg/kg and female mice of 218 mg/kg. Rats, rabbits acute percutaneous LD50> 2g/kg; rats acute inhalation LC50 (4 hours)> 5.65mg/L • air. (1 year) and 1 mg/(kg • d) in rats (2 years). No irritation to rabbit skin and eyes, no mutagenic effect, no skin allergies to guinea pigs. Low toxicity to birds, quail acute oral LD50 of 150mg/kg; LC50 of duck in 8 days feeding test is 1197mg/kg, while the value for quail is 816mg/kg. Fish poisoning LC50 (96 hours): carp 18.9mg/L, rainbow trout 19mg/L, blue gills 21mg/L. Bees LD50> 0.1mg/bee (contact),> 1mg/kg (oral). Daphnia LC50 (48 hours) was 26 mg/L.
Preparation
1-(4-chlorophenyl)-2-cyclopropyl-1-propanone was reacted with sodium hydride and methyl iodide to give 1-(4-chlorophenyl)-2-cyclopropyl-1-acetone, followed by reaction with CH3(CH2) 11S + (CH3)2CH3SO3-to produce ethylene oxide derivatives, and finally condensed with 1H-1, 2, 4-triazole, leading to the formation of Cyproconazole.
Chemical Properties
Colorless, crystalline solid. Odorless.
Uses
Cyproconazole is a fungicide treatment for wood preservative that prevents decay from fungi in above-ground applications.
Uses
Cyproconazole is used to inhibit demeythylation in fungal sterol synthesis. And it is used on greenhouse and field-grown roses and as a wood preservative, surface application or pressure treatment of wood for above-ground uses, including siding, plywood, millwork, shingles, lumber.
Uses
Cyproconazole is used as a systemic fungicide to control cereal and sugar beet foliar diseases: Septoria, rust, powdery mildew, Rhyncosporium, Cercospora and Ramularia. It is also used in fruit trees, vines, coffee, banana, turf, and vegetables to control Venturia, powdery mildew, rust, Mycosphaerela, Monilia, Mycena, Sclerotinia and Rhizoctonia.
Definition
ChEBI: 2-(4-chlorophenyl)-3-cyclopropyl-1-(1H-1,2,4-triazol-1-yl)butan-2-ol is a tertiary alcohol that is butan-2-ol substituted by a 4-chlorophenyl group at position 2, a cyclopropyl group at position 3 and a 1H-1,2,4-triazol-1-yl group at position 1. It is a member of monochlorobenzenes, a tertiary alcohol, a member of triazoles and a member of cyclopropanes.
Agricultural Uses
Fungicide: Used to control fungus on cereals, coffee beans; anthracnose and other diseases on turfgrass; used against rust and leaf spot disease.
Trade name
ALTO®; ALTO®, ELITE; ATEMI®; ATEMI-50-SL®;EVIPOL®; FLINT®; NOAH GOLD®; SAN-619 F®; SENTINEL®; SN 108266®
Potential Exposure
Triazole fungicide used to control fun- gus on cereals, coffee beans; anthracnose and other diseases on turfgrass; used against rust and leaf spot disease.
Metabolic pathway
Cyproconazole is a mixture of diastereiosomers and the preferential mammalian metabolism of one isomer has been observed. Pathways of metabolism in plants and mammals are quite similar. Metabolism primarily involves oxidation of the terminal methyl group to alcohols and carboxylic acids and/or hydroxylation at the 3-position. Loss of the alkyl cyclopropyl moiety or the triazolyl ring also occurs.
Shipping
UN3082 Environmentally hazardous substances, liquid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous haz- ardous material, Technical Name Required
Degradation
Cyproconazole is stable and decomposition was <5% after 2 years. It
was stable in aqueous solutions (at pH 1-9) for 35 days (50 °C) or 14
days (80 °C). It is slowly hydrolysed in hydrochloric acid or sodium
hydroxide.
14C-Labelled cyproconazole was applied at a rate equivalent to
100 g ha-1 to the surface of a 2 mm layer of loam soil. The samples were
exposed to natural sunlight for up to 72 days on open dishes or in flasks
fitted with plugs to trap volatile products. Samples were extracted at
intervals and analysed. After 37 days exposure on dishes, 50% of the
applied cyproconazole remained whereas 85% remained after 72 days in
the flasks. In the dishes, unextractable residues amounted to 13-20% of
the applied radiolabel after 20 days or more exposure, whereas in the
flasks it reached a maximum of 6% after 15 days. The products identified
in the dishes were: 1-(4-chlorophenyl)-2-(1H -1,2,4-triazol-l-yl)-
ethanone (2, 5%), 3-cyclopropyl-1-(1H-1,2,4-triazol-l-yl)butanone(4,4%),
2-(2- or 3-hydroxy-4-chlorophenyl)-3-cyclopropyl-1-(1H -1,2,4-triazol-1-yl)-
butan-2-ol (3 4%), and 4-chlorobenzoic acid 5 (Scheme 1) (PSD, 1991).
Degradation in the flasks was similar but occurred to a lesser extent. No
volatde products were trapped and recoveries of radiolabel were 94-
102%.
Incompatibilities
The triazoles are sensitive to heat, fric- tion, and impact. Sensitivity varies with the type substitu- tion to the triazole ring. Metal chelated and halogen substitution of the triazol ring make for a particularly heat sensitive material. Azido and nitro derivatives have been employed as high explosives. No matter the derivative these materials should be treated as explosives .
Waste Disposal
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Contact a licensed disposal facility about surplus and nonrecyclable solutions. Burn in a chemical incinerator equipped with an afterburner and scrubber. Extra care must be exercised as the material in an organic solvent is highly flammable. In accordance with 40CFR165, follow recom- mendations for the disposal of pesticides and pesticide con- tainers. Containers must be disposed of properly by following package label directions or by contacting your local or federal environmental control agency, or by con- tacting your regional EPA office. Incineration or permanga- nate oxidation.
Cyproconazole Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Qingdao Trust Agri Chemical Co.,Ltd | +8613573296305 | aroma@qdtrustagri.com | China | 299 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | sales@hbmojin.com | China | 12446 | 58 |
| Shaanxi Haibo Biotechnology Co., Ltd | +undefined18602966907 | qinhe02@xaltbio.com | China | 1000 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21667 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +8618949832763 | info@tnjchem.com | China | 2989 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29888 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 | alice@crovellbio.com | China | 8820 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 | linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 | sales@chemdad.com | China | 39916 | 58 |
Related articles
- Cyproconazole: a systemic triazole fungicide.
- Cyproconazole (CPZ) is a systemic triazole fungicide that controls rust, powdery mildew, and leaf spot caused by cotton aphids....
- Aug 23,2024
View Lastest Price from Cyproconazole manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-08-27 | Cyproconazole
94361-06-5
|
US $0.00 / kg | 100kg | 98 | 10000 | qingdao trust agri chemical co.,ltd | |
 |
2024-04-12 | Cyproconazole
94361-06-5
|
US $20.00-10.00 / kg | 1kg | 95% | 250000 Per Year | Shaanxi Haibo Biotechnology Co., Ltd | |
 |
2023-08-16 | Cyproconazole
94361-06-5
|
US $0.00 / KG | 1KG | 99% | 20tons | Hebei Mojin Biotechnology Co., Ltd |
-

- Cyproconazole
94361-06-5
- US $0.00 / kg
- 98
- qingdao trust agri chemical co.,ltd
-

- Cyproconazole
94361-06-5
- US $20.00-10.00 / kg
- 95%
- Shaanxi Haibo Biotechnology Co., Ltd
-

- Cyproconazole
94361-06-5
- US $0.00 / KG
- 99%
- Hebei Mojin Biotechnology Co., Ltd