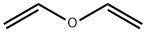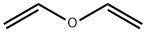Vinyl ether
- CAS No.
- 109-93-3
- Chemical Name:
- Vinyl ether
- Synonyms
- Vinydan;Vinethene;Vinyl ether;Vinesthesin;divinyl oxide;Divinyl ether;ethenyloxyethene;(vinyloxy)ethene;vinyloxyethylene;1,1'-oxybisethene
- CBNumber:
- CB4851587
- Molecular Formula:
- C4H6O
- Molecular Weight:
- 70.09
- MDL Number:
- MOL File:
- 109-93-3.mol
- MSDS File:
- SDS
| Melting point | -101.1°C |
|---|---|
| Boiling point | bp 28.4° |
| Density | d2020 0.774; d420 0.773 |
| refractive index | nD20 1.3989 |
| Water Solubility | 5.25g/L(37 ºC) |
| Dielectric constant | 3.9(20℃) |
| EWG's Food Scores | 1 |
| FDA UNII | 2H2T044E11 |
| ATC code | N01AA02 |
| EPA Substance Registry System | Ethene, 1,1'-oxybis- (109-93-3) |
Vinyl ether Chemical Properties,Uses,Production
Description
Divinyl ether is a mobile, colorless, inflammable liquid with a characteristic sweet odor, bp 28.4 ?C. Approximately 4 % ethanol is added as a stabilizer. Divinyl ether may contain 0.0077 % of a stabilizer, phenyl-α- naphthylamine, that produces a slightly reddish fluorescence.
Chemical Properties
Clear, colorless liquid with a characteristicodor; boils at 28.5°C (83°F); density 0.774[at 20°C (68°F)]; soluble in most organicsolvents, slightly soluble in water [0.53 g/dLat 37°C (98°F)].
Uses
Copolymer with 3–5% polyvinyl chloride for plastic products such as clear plastic bottles; medicine (anesthetic, for brief operations only).
Uses
Vinyl ether is used as an anesthetic and inorganic synthesis.
Indications
Divinyl ether is used for minor surgery, obstetrics, dentistry, and inducing anesthesia. A certain degree of hepatotoxicity rules out long-term use. Hypersecretion has also been observed. Divinyl ether increases and decreases in tissue levels faster than diethyl ether.
Definition
ChEBI: Vinyl ether is an ether.
General Description
A clear colorless liquid with a characteristic odor. Flash point below -22°F. Less dense than water. Autoignition temperature 680°F. Slightly soluble in water; miscible with alcohol, acetone, chloroform, and ether. Must be protected from light.Vapors heavier than air. Toxic by inhalation.
Air & Water Reactions
Highly flammable. Slightly soluble in water. Oxidizes readily in air to form unstable peroxides that may explode spontaneously [Bretherick, 1979 p.151-154, 164].
Reactivity Profile
Ethers, such as DIVINYL ETHER, can act as bases. They form salts with strong acids and addition complexes with Lewis acids. The complex between diethyl ether and boron trifluoride is an example. Ethers may react violently with strong oxidizing agents. In other reactions, which typically involve the breaking of the carbon-oxygen bond, ethers are relatively inert. Vinyl ether is miscible with alcohol, acetone, chloroform and ether and must be protected from light [Hawley].
Health Hazard
Vinyl ether exhibits low inhalation toxicity.Like ethyl ether, inhalation of its vapors pro duces an anesthetic effect. At concentrationsabove 5% (by volume in air), it can causeunconsciousness in humans. Exposure to a5.1% concentration of its vapor (in air) waslethal to mice.
Divinyl ether exhibited mutagenicity inmicrosomal assay and microbial mutationtests. There is no report on its carcinogenicityin animals or humans.
Health Hazard
Inhalation or contact with material may irritate or burn skin and eyes. Fire may produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.
Fire Hazard
Highly flammable; flash point (closed cup) <
-30°C (<-22°F); vapor density 2.4 (air =
1); the vapor is heavier than air and can travel
a considerable distance to a source of igni tion and flash back; autoignition temperature
360°C (680°F); can be ignited by lightning
or static electricity; fire-extinguishing agent:
dry chemical, foam, or CO2; use a water
spray to keep fire-exposed containers cool
and to disperse the vapors
Vinyl ether forms explosive mixtures with
air, with LEL and UEL values of 1.7%
and 27% by volume of air, respectively. It
forms unstable peroxides on long standing or
in the presence of oxygen. These peroxides
can explode spontaneously or when heated.
It decomposes to acetaldehyde (toxic and
highly flammable) when exposed to light.
Safety Profile
Mildly toxic by inhalation. Mutation data reported. Prolonged exposure causes liver injury. A very dangerous fire hazard when exposed to heat or flame; can react vigorously with oxidizing materials. A severe explosion hazard in the form of vapor when exposed to heat or flame. Forms peroxides when exposed to air or oxygen. Hypergolic reaction with concentrated nitric acid. To fight fire, use CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes. Used as an inhalation anesthetic. See also ETHERS.
Synthesis
Divinyl ether is prepared by the reaction of liquefied alkali hydroxide or sodium alkoxide on β,β_x0002_-dihalodiethyl ether or by pyrolysis of α,α_x0002_-dichlorodiethyl ether in the gas phase at 600 – 800 ?C in the presence of barium chloride catalyst .
storage
Vinyl ether is stored in a flammable-liquidsstorage room or cabinet separated from oxidizing and combustible materials. It shouldbe protected from physical damage andfrom static electricity or lightning. It isshipped in amber glass bottles or metaldrums.
Waste Disposal
Vinyl ether is mixed with an excess ofhigher-boiling solvent and burned in a chem ical incinerator equipped with an afterburnerand scrubber. Small amounts, free of peroxides, can be evaporated in a fume hood awayfrom a source of ignition.
Vinyl ether Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of2
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 | linda@hubeijusheng.com | CHINA | 22968 | 58 |
| career henan chemical co | +86-0371-86658258 15093356674; | factory@coreychem.com | China | 29826 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +86-89586680 +86-13289823923 | 1026@dideu.com | China | 9116 | 58 |
| Mainchem Co., Ltd. | +86-0592-6210733 | sale@mainchem.com | China | 32360 | 55 |
| Syntechem Co.,Ltd | info@syntechem.com | China | 12990 | 57 | |
| Shaanxi Dideu Medichem Co. Ltd | 029-81124267 15229202216 | 1073@dideu.com | China | 10011 | 58 |
| Hubei xinjing new material co. LTD | 027-83496456 15629169208 | hbxj20@xinjingchem.com | China | 2 | 58 |
View Lastest Price from Vinyl ether manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2020-01-13 | Vinyl ether
109-93-3
|
US $1.00 / KG | 1KG | 98%-99.9% | 100kg | Career Henan Chemical Co |
-

- Vinyl ether
109-93-3
- US $1.00 / KG
- 98%-99.9%
- Career Henan Chemical Co
109-93-3(Vinyl ether)Related Search:
1of4