TRIPARANOL
- CAS No.
- 78-41-1
- Chemical Name:
- TRIPARANOL
- Synonyms
- mer29;MER 2p;Clotrox;Diticyl;nsc65345;Drenaren;Metasqua;Acosterina;Metasclene;metasqualene
- CBNumber:
- CB5498433
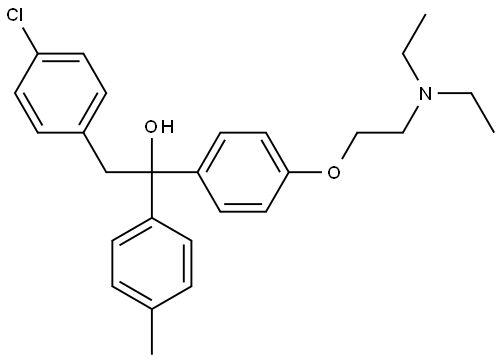
- Molecular Formula:
- C27H32ClNO2
- Molecular Weight:
- 438
- MDL Number:
- MFCD00865748
- MOL File:
- 78-41-1.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Triparanol ≥97% (HPLC), powder | T5200 | 5mg | $236 |
| Triparanol ≥97% (HPLC), powder | T5200 | 25mg | $860 |
| Triparanol ≥98% | 20918 | 500μg | $54 |
| Triparanol ≥98% | 20918 | 1mg | $94 |
| Triparanol ≥98% | 20918 | 5mg | $207 |
| Melting point | 102.9-103.7 °C |
|---|---|
| Density | 1.0173 (rough estimate) |
| refractive index | 1.5830 (estimate) |
| storage temp. | 2-8°C |
| solubility | DMSO: 15 mg/mL |
| Boiling point | 235-240 °C(Press: 0.08 Torr) |
| pka | 13.44±0.29(Predicted) |
| form | solid |
| color | off-white |
| InChI | 1S/C27H32ClNO2/c1-4-29(5-2)18-19-31-26-16-12-24(13-17-26)27(30,23-10-6-21(3)7-11-23)20-22-8-14-25(28)15-9-22/h6-17,30H,4-5,18-20H2,1-3H3 |
| InChIKey | SYHDSBBKRLVLFF-UHFFFAOYSA-N |
| SMILES | CCN(CC)CCOc1ccc(cc1)C(O)(Cc2ccc(Cl)cc2)c3ccc(C)cc3 |
| FDA UNII | 63S8C3RXGS |
| UNSPSC Code | 12352200 |
| NACRES | NA.77 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H318 | |||||||||
| Precautionary statements | P280-P301+P312+P330-P305+P351+P338+P310 | |||||||||
| PPE | dust mask type N95 (US), Eyeshields, Gloves | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 22-41 | |||||||||
| Safety Statements | 26-39 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | KK2400000 | |||||||||
| Storage Class | 11 - Combustible Solids | |||||||||
| Hazard Classifications | Acute Tox. 4 Oral Eye Dam. 1 |
|||||||||
| NFPA 704 |
|
TRIPARANOL price More Price(11)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | T5200 | Triparanol ≥97% (HPLC), powder | 78-41-1 | 5mg | $236 | 2026-03-19 | Buy |
| Sigma-Aldrich | T5200 | Triparanol ≥97% (HPLC), powder | 78-41-1 | 25mg | $860 | 2026-03-19 | Buy |
| Cayman Chemical | 20918 | Triparanol ≥98% | 78-41-1 | 500μg | $54 | 2024-03-01 | Buy |
| Cayman Chemical | 20918 | Triparanol ≥98% | 78-41-1 | 1mg | $94 | 2024-03-01 | Buy |
| Cayman Chemical | 20918 | Triparanol ≥98% | 78-41-1 | 5mg | $207 | 2024-03-01 | Buy |
TRIPARANOL Chemical Properties,Uses,Production
Originator
Mer-29,Merrell National,US,1960
Uses
Desmosterol Delta 24 (D24) reductase inhibitor
Uses
Triparanol has been used to inhibit cholesterol biosynthesis in lipid rafts1. Triparanol has also been used as a 3β-hydroxysterol-Δ24-reductase inhibitor to study its effect on delayed-rectifier potassium current (Iks) channels2.
Definition
ChEBI: Triparanol is a stilbenoid. It has a role as an anticoronaviral agent.
Manufacturing Process
4-(β-diethylaminoethoxy)-4-methylbenzophenone was prepared as follows: a
mixture of 200 g of 4-hydroxy-4-methylbenzophenone, 55 g of powdered
sodium methoxide and 400 ml of ethanol was stirred for 30 minutes. A
solution of 150 g of β-diethylaminoethyl chloride in 300 ml of toluene was
added and the mixture was refluxed four hours. The solvent was removed, the
residue was taken up in ether, extracted with 5% NaOH solution, twice with
water, the ether was removed and the residue was distilled. The product was
obtained as an oil boiling at 232°C at 0.6 mm.
1 liter of a 0.45 N ethereal solution of p-chlorobenzyl magnesium chloride was
added in 30 minutes to a stirred solution of 104 g (0.35 mol) of 4-(β-
diethylaminoethoxy)-4-methylbenzophenone in 400 ml of dry ether. After
stirring an additional hour, the mixture was decomposed by pouring onto 1
liter of cold 10% ammonium chloride solution, the ether solution was washed
with water, and the ether was replaced with hot isopropanol containing a trace
of ammonia. 1-[p-(β-diethylaminoethoxy)phenyl]-1-phenyl-2-p-tolyl-2-p_x0002_chloroethanol separated as white crystals, melting at 104% to 106°C.
Therapeutic Function
Antihyperlipidemic
Biochem/physiol Actions
Triparanol is known to repress Hedgehog signaling in cancer cells and can also inhibit tumor growth3.
TRIPARANOL Preparation Products And Raw materials
Raw materials
Preparation Products
TRIPARANOL Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| TargetMol Chemicals Inc. | +1-781-999-5354; +17819995354 | marketing@targetmol.com | United States | 32467 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-89586680 +86-18192503167 | 1026@dideu.com | China | 10000 | 58 |
| Hu Bei Jiutian Bio-medical Technology CO.,Ltd | 027-88013699 17354350817 | Ryan@jiutian-bio.com | China | 5945 | 58 |
| NANJING YINGWEN BIOTECHNOLOGY CO LTD | +8613382787392 | sales@aeechem.com | China | 10000 | 58 |
| J & K SCIENTIFIC LTD. | 18210857532; 18210857532 | jkinfo@jkchemical.com | China | 96815 | 76 |
| Chemsky (shanghai) International Co.,Ltd | 021-50135380 | shchemsky@sina.com | China | 15402 | 60 |
| Guangzhou Isun Pharmaceutical Co., Ltd | 020-39119399 18927568969 | isunpharm@qq.com | China | 4769 | 55 |
| Sigma-Aldrich | 021-61415566 800-8193336 | orderCN@merckgroup.com | China | 51395 | 80 |
| Shanghai EFE Biological Technology Co., Ltd. | 021-65675885 18964387627 | info@efebio.com | China | 9803 | 58 |
| Aikon International Limited | 025-58859352 19370895928 | sales01@aikonchem.com | China | 15082 | 58 |
View Lastest Price from TRIPARANOL manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-22 | Triparanol
78-41-1
|
US $678.00-298.00 / mg | 99.73% | 10g | TargetMol Chemicals Inc. |
-

- Triparanol
78-41-1
- US $678.00-298.00 / mg
- 99.73%
- TargetMol Chemicals Inc.