アストロマイシン 化学特性,用途語,生産方法
解説
C17H35N5O6(405.49).ホーチマイシンA(fortimicin A)ともいう.Micromonospora olivasterosporaが産生するアミノ配糖体抗生物質.白色の粉末.分解点200 ℃.[α]D25"+87.5°(水).グラム陽性球菌およびグラム陰性桿(かん)菌に有効な化学療法剤として臨床に使用されている.細菌のリボソームに作用し,タンパク質合成を阻害し,殺菌的に作用する.腸管からの吸収が悪く,筋肉注射または点滴静注する.腎毒性,聴覚毒性がアミノ配糖体抗生物質のなかでもっとも低い.β-ラクタム系,ほかのアミノ配糖体抗生物質と交差耐性を示さない.硫酸塩はLD50 380 mg/kg(マウス,静注)[CAS 55779-06-1][CAS 72275-67-3:二硫酸塩]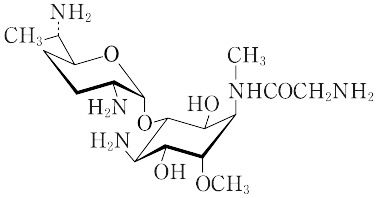 "
"
森北出版「化学辞典(第2版)
効能
抗生物質, タンパク質合成阻害薬
定義
ChEBI: An amino cyclitol glycoside that is L-chiro-inositol in which the hydroxy groups at positions 1, 4, and 6 are replaced by aminoacetyl)methylamino, amino, and methoxy groups, respectively, and in which the hydroxy group at posi
ion 3 is converted to the corresponding 2,6-diamino-2,3,4,6,7-pentadeoxy-beta-L-lyxo-heptopyranoside. The major component of fortimicin, obtained from Micromonospora olivasterospora. It is adm
nistered (as the sulfate salt) by intramuscular injection or intravenous infusion for the treatment of severe systemic infections due to sensitive Gram-negative organisms.
抗菌性
A pseudodisaccharide aminoglycoside produced by
Micromonospora olivoasterospora. Formulated as the sulfate.
Intrinsic activity is similar to that of amikacin for most
groups of organisms, but activity against Ps. aeruginosa is relatively
poor. It is resistant to many aminoglycoside-modifying
enzymes, but is sensitive to AAC(3) and the APH(2″)/AAC(6′)
bifunctional enzyme.
Peak concentrations of 10–12 mg/L were found in the
blood following 200 mg intravenous or intramuscular administration
to volunteers. The plasma half-life was 1.5–2 h. Over
85% of the drug was recovered in urine during the 8 h following
administration.
Toxicity and side effects are similar to those observed with
other aminoglycosides. Where the drug is available it is used
instead of amikacin in the treatment of infections caused by
susceptible organisms.
アストロマイシン 上流と下流の製品情報
原材料
準備製品