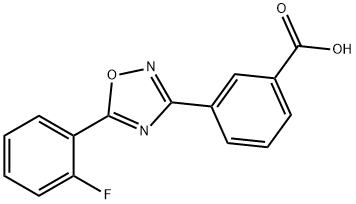
Ataluren
|
|
Ataluren 속성
- 녹는점
- 241 - 242°C
- 끓는 점
- 503.7±60.0 °C(Predicted)
- 밀도
- 1.379
- 저장 조건
- Refrigerator
- 용해도
- DMSO (약간 용해됨)
- 물리적 상태
- 흰색 내지 회백색 고체.
- 산도 계수 (pKa)
- 3.58±0.10(Predicted)
- 색상
- 흰색에서 황백색까지
- InChI
- InChI=1S/C15H9FN2O3/c16-12-7-2-1-6-11(12)14-17-13(18-21-14)9-4-3-5-10(8-9)15(19)20/h1-8H,(H,19,20)
- InChIKey
- OOUGLTULBSNHNF-UHFFFAOYSA-N
- SMILES
- C(O)(=O)C1=CC=CC(C2N=C(C3=CC=CC=C3F)ON=2)=C1
- CAS 데이터베이스
- 775304-57-9
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| HS 번호 | 2933998090 |
|---|
| 그림문자(GHS): |
 
|
|||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Warning | |||||||||||||||||||||
| 유해·위험 문구: |
|
|||||||||||||||||||||
| 예방조치문구: |
|
Ataluren C화학적 특성, 용도, 생산
개요
Ataluren is a drug marketed under the trade name Translarna® which was developed by PTC Therapeutics and approved by the European Union in May 2014 for the treatment of Duchenne’s muscular dystrophy (DMD) and potentially other genetic disorders. Ataluren renders ribosomes less sensitive to premature stop or ‘read-through’ codons, which are thought to be beneficial in diseases such as DMD and cystic fibrosis.용도
Nonsense mutations create a premature termination of mRNA translation and have been implicated in various genetic disorders, including muscular dystrophy and cystic fibrosis. PTC-124 is a nonaminoglycoside that has been reported to selectively induce ribosomes to read through premature nonsense stop signals on mRNA, thus allowing the production of full-length, functional proteins. In a mouse model of cystic fibrosis caused by nonsense mutations, PTC-124 treatment (60 mg/kg s.c. injection or 0.3-0.9 mg/ml orally) has been shown to restore cystic fibrosis transmembrane conductance regulator (CFTR) protein expression and function. The target activity of PTC-124 was initially evaluated by firefly luciferase reporter cell-based nonsense codon assay (IC50 = 7 nM); however, subsequent assessments using a Renilla reniformis luciferase reporter have failed to produce nonsense codon suppression activity. Thus, while PTC-124 is in clinical testing in patients with nonsense mutations within the CFTR or dystrophin genes, controversy surrounds its exact mechanism of action.[Cayman Chemical]Mechanism of action
The mechanism of action of Ataluren (PTC124) is to generate functionally normal myotonic dystrophy proteins by facilitating ribosomal read-through of nonsense mutations, thereby bypassing the pathogenic variant and continuing the translation process. Under normal conditions, ribosomes move along the mRNA that links amino acids into proteins until they reach the stop codon. When the ribosome encounters a premature termination codon (PTC) due to a nonsense mutation, eukaryotic release factors [eRF1 (green) and eRF3 (turquoise) complexed with GTP (yellow)] are recruited, and translation is terminated prematurely to produce the truncated protein. Ataluren is thought to interact with the ribosome to promote the recruitment of the proximity-recognition tRNAs, which in turn inhibits the nonsense mutation, allowing the PTC to be read through and synthesised. PTC to be read through and synthesise full-length proteins. The red amino acid on the near-recognition tRNA is incorporated into the read-through protein product. In the Ataluren-mediated nonsense repression model, the X on the tRNA indicates a mispairing at codon position three.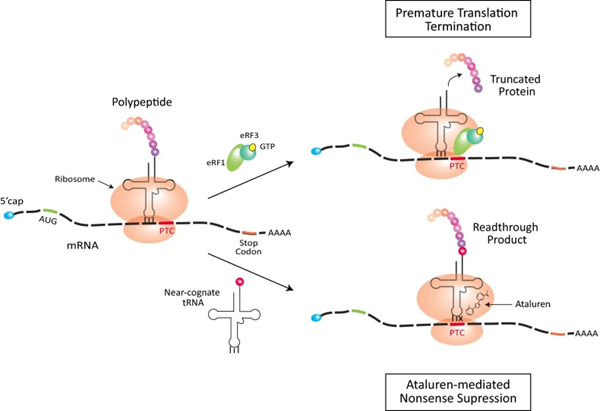
Synthesis
The sequence to construct ataluren, which was described by the authors at PTC Therapeutics, commenced with commercially available methyl 3-cyanobenzoate (38). This ester was exposed to hydroxylamine in aqueous tert-butanol and warmed gently until the reaction was deemed complete. Then this mixture was treated with 2-fluorobenzoyl chloride dropwise and subsequently triethylamine dropwise. To minimize exotherm and undesired side products, careful control of the addition of reagents was achieved through slow dropwise addition of these liquid reagents. Upon complete consumption of starting materials and formation of amidooxime 39, the aqueous reaction mixture was then heated to 85 ?? to facilitate 1,2,4-oxadiazole formation, resulting in the tricyclic ester 40 in excellent yield across the three steps. Finally, saponification of ester 40 through the use of sodium hydroxide followed by acidic quench gave ataluren (V) in 96% over the two-step sequence.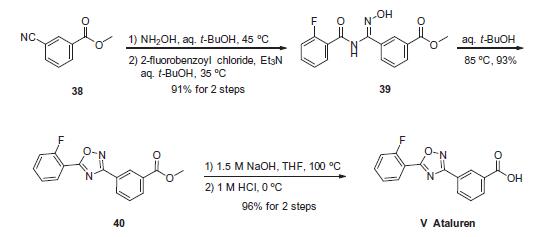
Ataluren 준비 용품 및 원자재
원자재
5-(2-fluorophenyl)-3-(3-methylphenyl)-1,2,4-oxadiazole
3-(Aminoiminomethyl)benzoic acid methyl ester
메틸3-시아노벤조에이트
벤조산,3-[5-(2-플루오로페닐)-1,2,4-옥사디아졸-3-일]-,메틸에스테르
3-[(하이드록시아미노)이미노메틸]-벤조산
3-메틸벤젠카복시이미드아마이드염산염
2-플루오로벤조산
3-시아노벤조익산
플로로(2-)벤조일
준비 용품
Ataluren 공급 업체
글로벌( 226)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Shanghai Famo Biotech Co Ltd | +86-36680037 +86-18550473860 |
mayan@famobiotech.com;sales@famobiotech.com | China | 528 | 58 |
| shandong perfect biotechnology co.ltd | +86-53169958659; +8618596095638 |
sales@sdperfect.com | China | 294 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Hangzhou ICH Biofarm Co., Ltd | +86-0571-28186870; +undefined8613073685410 |
sales@ichemie.com | China | 990 | 58 |
| Anhui Ruihan Technology Co., Ltd | +8617756083858 |
daisy@anhuiruihan.com | China | 994 | 58 |
| HEBEI SHENGSUAN CHEMICAL INDUSTRY CO.,LTD | +8615383190639 |
admin@86-ss.com | China | 1000 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29798 | 60 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32836 | 60 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29888 | 58 |
| Biochempartner | 0086-13720134139 |
candy@biochempartner.com | CHINA | 967 | 58 |
Ataluren 관련 검색:
아세트산 1-((비스(4-플루오로페닐)메틸실릴)메틸)-1H-1,2,4-트리아졸 엽산 디페닐-4-카본산 테트라코나졸 플루퀸코나졸
PTC-209 (hydrobroMide)
PTC-209 (PTC209
1-[[(2S,3S)-3-(2-Chlorophenyl)-2-(4-fluorophenyl)oxiran-2-yl]methyl]-1,2,4-triazole
3-(Trifluoromethyl)benzoic acid
3-(5-METHYL-1,2,4-OXADIAZOL-3-YL)BENZOIC ACID
Ataluren (PTC124)
3-(5-METHYL-1,2,4-OXADIAZOL-3-YL)BENZALDEHYDE
3-(5-PHENYL-1,2,4-OXADIAZOL-3-YL)BENZOIC ACID
[3-(5-METHYL-1,2,4-OXADIAZOL-3-YL)PHENYL]METHANOL
AQUANTRAAL
BENZOICACID
Ataluren-D4