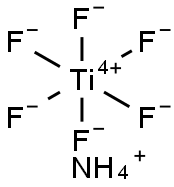헥사플루오로티탄산 암모늄
|
|
헥사플루오로티탄산 암모늄 속성
- 녹는점
- 1660 °C(lit.)
- 끓는 점
- 3287 °C(lit.)
- 밀도
- 4.5 g/mL at 25 °C(lit.)
- 용해도
- H2O: 약간 용해됨(lit.)
- 물리적 상태
- 철사
- 색상
- 하얀색
- 수소이온지수(pH)
- pH(50g/l, 25℃) : 2.0~4.0
- 수용성
- 물에 불용성.
- Hydrolytic Sensitivity
- 0: forms stable aqueous solutions
- 노출 한도
- ACGIH: TWA 2.5 mg/m3
NIOSH: IDLH 250 mg/m3
- CAS 데이터베이스
- 16962-40-6(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
헥사플루오로티탄산 암모늄 C화학적 특성, 용도, 생산
화학적 성질
white crystalline powder, crystals and/or chunks용도
Ammonium hexafluorotitanate is used as an anti-corrosive cleaning agent. It is also used for the production of ceramics and glass. Further, it is used in the preparation of synthetic gems.Safety Profile
Poison by intravenous route. See also FLUORIDES, AMMONIA, and TITANIUM COMPOUNDS. When heated to decomposition it emits very toxic fumes of Fand NOx,.헥사플루오로티탄산 암모늄 준비 용품 및 원자재
원자재
준비 용품
헥사플루오로티탄산 암모늄 공급 업체
글로벌( 267)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 |
zheyansh@163.com | CHINA | 3620 | 58 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 |
sales@chemdad.com | China | 39916 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49390 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-029-81138252 +86-18789408387 |
1057@dideu.com | China | 3537 | 58 |
헥사플루오로티탄산 암모늄 관련 검색:
염화티탄(Ⅳ) 헥사플루오로티탄산 암모늄 티타늄(IV) 이소프로폭시드 테트라-n-부톡시티탄 티탄산 납 비스(2-프로판올에이토)비스(2,4-펜탄디오네이토)티타늄 바륨 티타네이트(IV) 디칼륨 헥사플루오로티타늄산염 티타노센 디염화물 아나타제 테트라프로필오르토티탄산염 브롬화 티타늄(IV) 티타늄 타이타늄산 칼륨(티탄산 칼륨) 1-뷰탄올, 타이타늄(4+) 염, 호모중합체(1-부탄올, 티타늄(4+) 염, 호모중합체) 산화 스트론슘 티타늄 옥소(29H, 31H프탈로사이아닌아토(2-)-N29,N30,N31, N32),(SP-5-12)티타늄 포타슘 타이타늄 옥살레이트