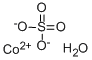황산 코발트
|
|
황산 코발트 속성
- 녹는점
- decomposes at 1140℃ [JAN85]
- 밀도
- d425 3.71
- 증기압
- 0Pa at 20℃
- 용해도
- H2O: 용해성
- 물리적 상태
- 붉은 사방정계 결정
- 색상
- red orthorhombic crystals, crystalline
- 수용성
- 끓는 H2O [MER06]에 천천히 용해됩니다. g/100g 용액 H2O: 19.7 ± 0.1(0°C), 27.2 ± 0.1(25°C), 27.8(100°C); 고체상, CoSO4 · 7H2O (0°C, 25°C), CoSO4 ·H2O (100°C) [KRU93]
- Merck
- 13,2473
- InChI
- InChI=1S/Co.H2O4S.H2O/c;1-5(2,3)4;/h;(H2,1,2,3,4);1H2/q+2;;/p-2
- InChIKey
- BGORGFZEVHFAQU-UHFFFAOYSA-L
- SMILES
- S([O-])([O-])(=O)=O.O.[Co+2]
- LogP
- -1.031 (est)
- CAS 데이터베이스
- 10124-43-3(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,N | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 49-42/43-51/53-50/53-22-68-60 | ||
| 안전지침서 | 53-23-36/37-45-61-60-22 | ||
| 유엔번호(UN No.) | UN 3082 9/PG 3 | ||
| WGK 독일 | 2 | ||
| 유해 물질 데이터 | 10124-43-3(Hazardous Substances Data) | ||
| 기존화학 물질 | KE-06128 | ||
| 중점관리물질 필터링 | 별표1-149 |
| 그림문자(GHS): |
  
|
|||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 신호 어: | Danger | |||||||||||||||||||||||||||||||||||||||||||||||||
| 유해·위험 문구: |
|
|||||||||||||||||||||||||||||||||||||||||||||||||
| 예방조치문구: |
|
황산 코발트 C화학적 특성, 용도, 생산
개요
The blue, crystalline hydrate Co2(SO4)3.18H2O is prepared by the oxidation of cobalt(II) sulphate in 8N sulphuric acid either electrolytically or chemically with ozone or fluorine. It is stable in the dry state, but is decomposed by water with evolution of oxygen; it is fairly stable in solution in dilute sulphuric acid. Cobalt(III) alums MCo(SO4)2.12H2O (M = K, Rb, Cs or NH4) can be isolated as blue crystals from the mixed cooler solutions of the two sulphates in dilute sulphuric acid. The potassium alum is diamagnetic, the rubidium salt has a magnetic moment less than 1 B.M. and the ammonium alum has a moment of 2.1 B.M. at 304°K. The hydrated sulphate also has a small positive magnetic susceptibility. The sulphate is believed like the alums to contain the [Co(H2O)6]3+ ion.화학적 성질
Red powder or rose pink crystalline solid. Odorless.용도
Ceramics, pigments, glazes, in plating baths for cobalt, additive to soils, catalyst, paint and ink drier, storage batteries.일반 설명
Odorless rose-pink solid. Sinks and mixes with water.공기와 물의 반응
Water soluble.반응 프로필
Acidic salts, such as Cobalt sulfate , are generally soluble in water. The resulting solutions contain moderate concentrations of hydrogen ions and have pH's of less than 7.0. They react as acids to neutralize bases. These neutralizations generate heat, but less or far less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. They usually do not react as either oxidizing agents or reducing agents but such behavior is not impossible. Many of these compounds catalyze organic reactions.위험도
May not be used in food products (FDA). Possible carcinogen.건강위험
Inhalation causes shortness of breath and coughing; permanent disability may occur. Ingestion causes pain and vomiting. Contact with eyes or skin causes irritation.화재위험
Special Hazards of Combustion Products: Toxic cobalt oxide fumes may form in fire.Safety Profile
Poison by intravenous and intraperitoneal routes. Moderately toxic by ingestion. Questionable carcinogen. When heated to decomposition it emits toxic fumes of SOx See also COBALT COMPOUNDS.잠재적 노출
Many be used to catalyze organic reactions.Carcinogenicity
Cobalt sulfate is reasonably anticipated to be a human carcinogenbased on sufficient evidence of carcinogenicity from studies in experimental animals.운송 방법
UN3288 Toxic solids, inorganic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required.Purification Methods
Crystallise it three times from conductivity water (1.3mL/g) between 100o and 0o depending on which hydrate is required. The heptahydrate crystallises below 44o and is efflorescent with m 97o . Between 44o and 70o the monoclinic hexahydrate CoSO4.6H2O m 41.5o is formed, and above 70o the monohydrate CoSO4.H2O m 71o is obtained. The pale reddish or lavender-coloured anhydrous salt is obtained by heating the hydrate above 250o, boiling with conc H2SO4 or heating with (NH4)2SO4).비 호환성
Aqueous solution reacts with bases, generating some heat. May react as either oxidizing agents or reducing agents폐기물 처리
Use a licensed professional waste disposal service to dispose of this material. All federal, state, and local environmental regulations must be observed.황산 코발트 준비 용품 및 원자재
원자재
준비 용품
황산 코발트 공급 업체
글로벌( 231)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| henan kanbei chemical co.,ltd | +undefined-86-1523780-4566 +undefined15237804566 |
henankanbeichemical@163.com | China | 511 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 |
sales@hbmojin.com | China | 12446 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 |
peter@yan-xi.com | China | 5873 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21667 | 55 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418679 +8618949832763 |
info@tnjchem.com | China | 2989 | 55 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 |
linda@hubeijusheng.com | CHINA | 28180 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 |
linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Shandong chuangyingchemical Co., Ltd. | 18853181302 |
sale@chuangyingchem.com | CHINA | 5909 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39916 | 58 |
황산 코발트 관련 검색:
황산 나트륨 황산코발트 디메틸설페이트 황산바륨 황산카나마이신 황산마그네슘(7수화물) 코발트분 황산알루미늄 황산암모늄 황산 망간 질산 코발트 코발트 암모늄 황산염 황산 코발트
Cobalt sulfate
Cobalt(Ⅱ) sulfate hydrate,COBALT SULFATE HYDRATE,COBALT SULFATE MONOHYDRATE,Cobalt(Ⅱ) sulfate anhydrous
sulfate
Ammonium cobalt(II) sulfate hexahydrate
COBALT SULFATE HYDRATE,COBALT SULFATE MONOHYDRATE