|
|
| | N-BOC-(R)-2-AMINO-3-HYDROXY-3-METHYLBUTANOIC ACID Basic information |
| Product Name: | N-BOC-(R)-2-AMINO-3-HYDROXY-3-METHYLBUTANOIC ACID | | Synonyms: | BOC-(R)-2-AMINO-3-HYDROXY-3-METHYLBUTANOIC ACID;(R)-Boc-2-aMino-3-hydroxy-3-Methyl-butyric acid;(R)-Boc-beta-hydroxy-valine;N-BOC-(R)-2-AMINO-3-HYDROXY-3-METHYLBUTANOIC ACID;Boc-(R)-2-amino-3-methylbutanoic acid;(R)-Boc-beta, beta-dimethyl-serine;N-Boc-3-hydroxy-D-valine, 97%;3-Hydroxy-D-valine, N-BOC protected | | CAS: | 288159-40-0 | | MF: | C10H19NO5 | | MW: | 233.26 | | EINECS: | | | Product Categories: | unnatural amino acids | | Mol File: | 288159-40-0.mol | 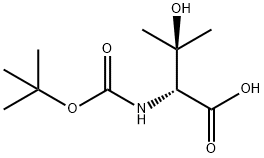 |
| | N-BOC-(R)-2-AMINO-3-HYDROXY-3-METHYLBUTANOIC ACID Chemical Properties |
| Boiling point | 391.1±37.0 °C(Predicted) | | density | 1.175±0.06 g/cm3(Predicted) | | storage temp. | 2-8°C | | pka | 3.62±0.10(Predicted) | | InChI | InChI=1S/C10H19NO5/c1-9(2,3)16-8(14)11-6(7(12)13)10(4,5)15/h6,15H,1-5H3,(H,11,14)(H,12,13)/t6-/m0/s1 | | InChIKey | SZVRVSZFEDIMFM-LURJTMIESA-N | | SMILES | C(O)(=O)[C@@H]([C@@](C)(O)C)NC(OC(C)(C)C)=O | | CAS DataBase Reference | 288159-40-0 |
| HazardClass | IRRITANT | | HS Code | 29225090 |
| | N-BOC-(R)-2-AMINO-3-HYDROXY-3-METHYLBUTANOIC ACID Usage And Synthesis |
| Chemical Properties | White powder | | Synthesis | General procedure for the synthesis of (R)-2-((tert-butoxycarbonyl)amino)-3-hydroxy-3-methylbutanoic acid from tert-butyl (S)-(1,3-dihydroxy-3-methylbutan-2-yl)carbamate: [Step 3] Synthesis of (R)-2-((tert-butoxycarbonyl)amino)-3-hydroxy-3-methylbutanoic acid: at -35 °C, an aqueous solution of sodium hypochlorite (5.0 mL, containing 0.8 g, 0.64 mmol) and aqueous sodium hypochlorite (10 mL, containing 2.4 g, 27 mmol) were added simultaneously and slowly to an acetonitrile containing Reference Example Compound 36 (2.8 g, 13 mmol), 2,2,6,6-tetramethylpiperidinium 1-yloxy (0.40 g, 2.6 mmol), and a standard neutral phosphate buffer (45 mL) (50 mL) solution. The reaction solution was stirred at -35 °C for 3 h. Acetic acid (1.0 mL) was added. Stirring was continued at -35°C for 3 hours, followed by the addition of water and extraction with ethyl acetate. The organic layer was washed sequentially with water and saturated aqueous sodium chloride, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The resulting crude product was dissolved in saturated aqueous sodium bicarbonate solution and washed with ethyl acetate. The aqueous layer was acidified with 3N hydrochloric acid and extracted again with ethyl acetate. The organic layer was washed with water and saturated aqueous sodium chloride, dried over anhydrous sodium sulfate and concentrated under reduced pressure to give 2.5 g (84% yield) of (R)-2-((tert-butoxycarbonyl)amino)-3-hydroxy-3-methylbutanoic acid (hereinafter referred to as Reference Example Compound 37). | | References | [1] Journal of Organic Chemistry, 2003, vol. 68, # 1, p. 177 - 179
[2] Nature Chemistry, 2010, vol. 2, # 4, p. 280 - 285
[3] Patent: EP2832724, 2015, A1. Location in patent: Paragraph 0188 |
| | N-BOC-(R)-2-AMINO-3-HYDROXY-3-METHYLBUTANOIC ACID Preparation Products And Raw materials |
|