|
|
| | BENZIODARONE Basic information |
| Product Name: | BENZIODARONE | | Synonyms: | BENZIODARONE;2-Ethyl-3-benzofuranyl 4-hydroxy-3,5-diiodophenyl ketone;Vasilar;2329labaz;2-ethyl-3-(3’,5’-diiodo-4’-hydroxybenzoyl)-cumarone;3-(3,5-diiodo-4-hydroxybenzoyl)-2-ethyl-benzofura;3,5-diiodo-4-hydroxyphenyl2-ethyl-3-benzofuranylketone;aethyl-2-(3’,5’-dijod-4’-oxybenzoyl)-3cumaron | | CAS: | 68-90-6 | | MF: | C17H12I2O3 | | MW: | 518.08 | | EINECS: | 200-695-2 | | Product Categories: | Aromatics;Heterocycles;Intermediates & Fine Chemicals;Pharmaceuticals | | Mol File: | 68-90-6.mol | 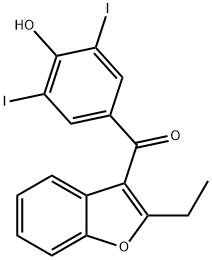 |
| | BENZIODARONE Chemical Properties |
| Melting point | 167° | | density | 1.9173 (estimate) | | storage temp. | Refrigerator | | solubility | Chloroform (Slightly), DMSO (Slightly), Ethyl Acetate (Slightly) | | form | Solid | | color | Beige | | Water Solubility | 5.881mg/L(20 ºC) | | CAS DataBase Reference | 68-90-6 |
| | BENZIODARONE Usage And Synthesis |
| Originator | Amplivix,Sigma-Tau | | Uses | Benziodarone is a uricosuric drug,it is used to effectively to control hyperuricemia in renal transplantation. | | Definition | ChEBI: Benziodarone is an aromatic ketone. | | Manufacturing Process | The starting product 2-ethyl-3-benzofuranyl p-hydroxyphenyl ketone
(benzaron) was prepared in 4 steps:
1. First step was a reaction of salicylic aldehyde with chloroacetone to produce
2-acetyl-1-benzofuran.
2. It was reduced by hydrazine hydrate in an alkaline medium by process of
Hyuang-Minlon, J.A.C.S., 1946, 68, 2487 to give 2-ethyl-1-benzofurane.
3. 2-Ethyl-3-(4-methoxybenzoyl)-1-benzofuran was obtained from 2-ethyl-1-
benzofuran and 4-methoxy-benzoylchloride in a presence of tin tetrachloride.
4. It was heated with pyridine hydrochloride at 200°-220°C to give 2-ethyl-3-
benzofuranyl p-hydroxyphenyl ketone (benzaron) as described in N.P.B.Hoi,
C.Beaudet; US Patent No. 3,012,042; Dec. 5, 1961.
Benzaron (1 part) was dissolved in a very slight excess of 3% caustic soda. To
this solution is gradually added a slight excess of iodine dissolved in a 25%
aqueous solution of potassium iodide. The resultant solution is acidified with a
20% solution of sodium bisulphite, centrifuged, washed with water and then
recrystallized in acetic acid to give of 2-ethyl-3-benzofuranyl 4-hydroxy-3,5-
diiodophenyl ketone. MP: 167°C. | | Therapeutic Function | Coronary vasodilator, Uricosuric | | Safety Profile | A poison bj7
intraperitoneal and intravenous routes.
Moderately toxic by ingestion. A flammable
liquid. When heated to decomposition it
emits toxic vapors of I-. |
| | BENZIODARONE Preparation Products And Raw materials |
|