- (+)-10-CAMPHORSULFONIMINE
-

- $9.80 / 1.79999995231628KG
-
2020-01-14
- CAS:107869-45-4
- Min. Order: 1g
- Purity: ≥99%
- Supply Ability: 100kg
|
| | (+)-10-CAMPHORSULFONIMINE Basic information |
| Product Name: | (+)-10-CAMPHORSULFONIMINE | | Synonyms: | (1R)-(+)-CAMPHORSULFONYLIMINE;(1R)-(+)-(10-CAMPHORSULFONYL)IMINE;(3aR,6S)-8,8-Dimethyl-4,5,6,7-tetrahydro-3H-3a,6-methanobenzo[c]isothiazole 2,2-dioxide;(1R)-(+)-10,10-Dimethyl-3-thia-4-azatricyclo[5.2.1.01,5]dec-4-ene 3,3-dioxide;(1R)-(+)-Camphorsulfonylimine, (3aR)-(+)-4,5,6,7-Tetrahydro-8,8-dimethyl-3H-3a,6-methano-2,1-benzisothiazole-2,2-dioxide;(3aR,6S)-4,5,6,7-Tetrahydro-8,8-dimethyl-3H-3a,6-methano-2,1-benzisothiazole 2,2-dioxide;(7R)-(+)-10,10-Dimethyl-3-thia-4-azatricyclo[5.2.1.0(1,5)]dec-4-ene 3,3-Dioxide
(3aR)-(+)-4,5,6,7-Tetrahydro-8,8-dimethyl-3H-3a,6-methano-2,1-benzisothiazole 2,2-Dioxide;(3AR)-(+)-4,5,6,7-TETRAHYDRO-8,8-DIMETHYL-3H-3A,6-METHANO-2,1-BENZISOTHIAZOLE 2,2-DIOXIDE | | CAS: | 107869-45-4 | | MF: | C10H15NO2S | | MW: | 213.3 | | EINECS: | 621-464-6 | | Product Categories: | Chiral Reagents;Bicyclic Monoterpenes;Biochemistry;Terpenes;Intermediates & Fine Chemicals;Pharmaceuticals | | Mol File: | 107869-45-4.mol | 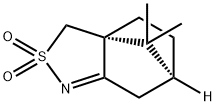 |
| | (+)-10-CAMPHORSULFONIMINE Chemical Properties |
| Melting point | 229-230 °C(lit.) | | Boiling point | 337℃ | | density | 1.50 | | refractive index | 31 ° (C=2, CHCl3) | | Fp | 158℃ | | storage temp. | Sealed in dry,Room Temperature | | solubility | Acetone, Dichloromethane, Hot Methanol | | form | Solid | | pka | -1.63±0.40(Predicted) | | color | White Cyrstalline | | InChI | InChI=1S/C10H15NO2S/c1-9(2)7-3-4-10(9)6-14(12,13)11-8(10)5-7/h7H,3-6H2,1-2H3/t7-,10-/m0/s1 | | InChIKey | ZAHOEBNYVSWBBW-XVKPBYJWSA-N | | SMILES | N1=C2[C@@]3(C(C)(C)[C@]([H])(C2)CC3)CS1(=O)=O | | CAS DataBase Reference | 107869-45-4 |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-36 | | WGK Germany | 3 | | HS Code | 2934.99.9001 |
| | (+)-10-CAMPHORSULFONIMINE Usage And Synthesis |
| Chemical Properties | White Cyrstalline Solid | | Uses | (+)-10-CAMPHORSULFONIMINE is a useful synthetic intermediate,it is used for asymmetric hydroxylation. | | Uses | A useful synthetic intermediate. Used for asymmetric hydroxylation. | | Synthesis | Example 1; Synthesis of (1R)-(+)-2,10-camphorsulfonamide (Scheme 3,3-1): Thionyl chloride (0.896 kg, 7.5 mol) was slowly added to a solution of (-)-camphorsulfonic acid (1.45 kg, 6.25 mol) in chloroform (7 L) over a period of 1 hour under refluxing conditions. The reaction mixture was refluxed for 16 h and then cooled to 4 °C using an ice bath. The cooled reaction mixture was slowly added to concentrated NH4OH (15 L) while controlling the rate of addition to keep the temperature below 15 °C. After the addition was completed, the mixture was stirred at room temperature for 4 hours. The organic layer was separated and the aqueous layer was extracted with chloroform (2 x 2 L). The combined organic extracts were washed with brine (4 L) and dried over MgSO4. After filtration, the filtrate was concentrated in vacuum and dried to give 1.2 kg (83% yield) of camphorsulfonamide. In another experiment, a comparable yield was obtained using 145 g scale of camphorsulfonamide with dichloromethane replacing chloroform for reaction and extraction. To a suspension of camphorsulfonamide (1.2 kg, 5.2 mol) in toluene (15 L) was added Amberlyst H+ resin (150 g), and the mixture was heated and refluxed for 4 hr while the resulting water was removed by azeotropic boiling using a Dean-Stark water separator. The reaction mixture was hot filtered to remove the resin. Cooling of the filtrate yielded a white solid, which was collected by filtration to give 1.02 kg (92% yield) of the target imine. To an ethanol (0.75 L) solution of the above imine (100 g, 0.47 mol) was added Raney nickel (100 g) and the mixture was hydrogenated at 40 psi for 2 hours. The catalyst was removed by filtration and the filtrate was concentrated in vacuum to give product 3-1 as a white solid. | | References | [1] Tetrahedron, 1986, vol. 42, # 14, p. 4035 - 4044
[2] Chemical Communications, 1998, # 15, p. 1549 - 1550
[3] Journal of the American Chemical Society, 1988, vol. 110, p. 8477
[4] Patent: US2006/128789, 2006, A1. Location in patent: Page/Page column 7
[5] Organic Process Research and Development, 2005, vol. 9, # 2, p. 193 - 197 |
| | (+)-10-CAMPHORSULFONIMINE Preparation Products And Raw materials |
|