BOC-11-AMINOUNDECANOIC ACID manufacturers
|
| | BOC-11-AMINOUNDECANOIC ACID Basic information |
| Product Name: | BOC-11-AMINOUNDECANOIC ACID | | Synonyms: | BOC-11-AMINOUNDECANOIC ACID;BOC-11-AMINOUNDECYLIC ACID;BOC-11-AUN-OH;BOC-NH-(CH2)10-COOH;11-(BOC-AMINO)-UNDECANOIC ACID;T-BUTOXYCARBONYL-11-AMINOUNDECANOIC ACID;T-BUTOXYCARBONYL-11-AMINOUNDECYLIC ACID;N-TERT-BUTYLOXYCARBONYL-11-AMINO-UNDECANOIC ACID | | CAS: | 10436-25-6 | | MF: | C16H31NO4 | | MW: | 301.42 | | EINECS: | | | Product Categories: | Amino Acids | | Mol File: | 10436-25-6.mol | 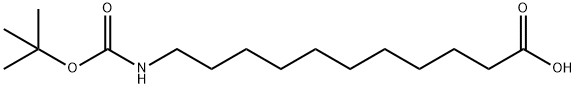 |
| | BOC-11-AMINOUNDECANOIC ACID Chemical Properties |
| Melting point | 67-68 °C | | Boiling point | 441.9±18.0 °C(Predicted) | | density | 1.006±0.06 g/cm3(Predicted) | | storage temp. | Sealed in dry,Room Temperature | | solubility | soluble in Methanol | | form | powder to crystal | | pka | 4.78±0.10(Predicted) | | color | White to Almost white | | BRN | 1975656 | | Major Application | peptide synthesis | | InChI | InChI=1S/C16H31NO4/c1-16(2,3)21-15(20)17-13-11-9-7-5-4-6-8-10-12-14(18)19/h4-13H2,1-3H3,(H,17,20)(H,18,19) | | InChIKey | HPTPZJBSQUULAV-UHFFFAOYSA-N | | SMILES | C(O)(=O)CCCCCCCCCCNC(OC(C)(C)C)=O | | CAS DataBase Reference | 10436-25-6(CAS DataBase Reference) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 22-24/25 | | WGK Germany | 3 | | HS Code | 2924.19.8000 | | HazardClass | IRRITANT | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | BOC-11-AMINOUNDECANOIC ACID Usage And Synthesis |
| Description | Boc-11-aminoundecanoic acid is an alkane chain with terminal carboxlic acid and Boc-protected amino groups. The compound can be used as a PROTAC linker in the synthesis of PROTACs. The terminal carboxylic acid can react with primary amine groups in the presence of activators (e.g. EDC, or HATU) to form a stable amide bond. The Boc group can be deprotected under mild acidic conditions to form the free amine. | | Chemical Properties | White powder | | Uses | Boc-11-aminoundecanoic acid is an Alkyl/ether-basedPROTAC linkercan be used in the synthesis of MS432 (HY-130602)[1]. | | reaction suitability | reaction type: Boc solid-phase peptide synthesis | | Synthesis | General procedure for the synthesis of Boc-11-aminoundecanoic acid from 11-aminoundecanoic acid and di-tert-butyl dicarbonate: 160 g of 11-aminoundecanoic acid, 2 liters of dioxane, 1.3 liters of distilled water, 208 g of sodium carbonate, and 173 g of di-tert-butyl dicarbonate were added sequentially to a 4-liter three-necked flask in a unit equipped with a mechanical stirrer and reflux condenser. The reaction mixture was heated to boiling point and the reaction was continued with stirring for 16 hours to obtain a clarified solution. Upon completion of the reaction, the mixture was cooled to 20 °C and subsequently poured into 800 g of ice and the pH was adjusted to 3-4 with 4 N hydrochloric acid, at which point a white precipitate was produced. The precipitate was separated by filtration, washed with 300 cm3 of water and then dried under reduced pressure at 20 °C (20 mm Hg, 2.7 kPa) to give 232 g of N-(tert-butoxycarbonyl)-11-aminoundecanoic acid in 95% yield. The melting point of the product (68°C) is in agreement with that reported in the literature J. Org. Chem. 1997,1987.Chem., 41,1350 (1976). | | IC 50 | Alkyl/ether | | References | [1] Wei J, et al.Discovery of a First-in-Class Mitogen-Activated Protein Kinase Kinase 1/2 Degrader.J Med Chem. 2019 Dec 12;62(23):10897-10911. DOI:10.1021/acs.jmedchem.9b01528 |
| | BOC-11-AMINOUNDECANOIC ACID Preparation Products And Raw materials |
|