|
|
| | Benzamidine hydrochloride Basic information |
| | Benzamidine hydrochloride Chemical Properties |
| Melting point | 86-88 °C(lit.) | | storage temp. | 2-8°C | | solubility | DMF: 25 mg/ml
DMSO: 25 mg/ml
Ethanol: 10 mg/ml
PBS (pH 7.2): 3 mg/ml | | form | Powder | | color | White to off-white | | PH | 4.0 to 6.5(50g/L, 25 ℃) | | Water Solubility | soluble | | BRN | 3594959 | | Stability: | Stable for 2 years from date of purchase as supplied. Solutions are not stable and must be prepared fresh daily | | InChI | InChI=1S/C7H8N2.ClH/c8-7(9)6-4-2-1-3-5-6;/h1-5H,(H3,8,9);1H | | InChIKey | LZCZIHQBSCVGRD-UHFFFAOYSA-N | | SMILES | C1(C(=N)N)=CC=CC=C1.Cl | | CAS DataBase Reference | 1670-14-0(CAS DataBase Reference) | | EPA Substance Registry System | Benzenecarboximidamide, monohydrochloride (1670-14-0) |
| Hazard Codes | Xi | | Risk Statements | 36/37/38 | | Safety Statements | 26-37/39-36 | | WGK Germany | 3 | | RTECS | CV6260000 | | F | 3-21 | | TSCA | TSCA listed | | HS Code | 29252900 | | Storage Class | 11 - Combustible Solids |
| | Benzamidine hydrochloride Usage And Synthesis |
| Description | Benzamidine hydrochloride is a reversible inhibitor of trypsin, trypsin-like enzymes, and serine proteases. A concentration of approximately 1 mM is used for general protease inhibition. To inhibit proteases from yeast, a range of 0.5 to 4.0 mM is used and it is for the most part interchangeable with pepstatin A.
In addition to being a strong competitive inhibitor of trypsin, benzamidine HCl has been also shown to be a strong competitive inhibitor of thrombin and plasmin. It was also found to be as effective as aprotinin in the prevention of glucagon degradation in human plasma. | | Chemical Properties | white to off-white powder | | Uses | Benzamidine is a reversible inhibitor of serine proteases, including trypsin, plasmin, and thrombin (Kis = 35, 350, and 220 μM, respectively). In addition to its use as a general serine protease inhibitor, benzamidine is used, when immobilized, to purify novel proteases. | | Preparation | To a stainless steel rocking autoclave equipped with a stirrer is added 103.0 gm (1.0 mole) of benzonitrile, 214.0 gm (4.0 mole) of ammonium chloride, and 306.0 gm (18.0 mole) of ammonia is introduced by means of a transfer bomb. The reaction mixture is heated at 150°C for 18 hr (pressure: 1300-6500 psig), cooled, and, with appropriate precautions for the safe control of the excess ammonia, is vented to atmospheric pressure. The reaction mixture is extracted with ether to remove approximately 5% unreacted benzonitrile, and then extracted with hot acetonitrile or ethanol to separate the amidine hydrochloride from the unreacted ammonium chloride. Concentration of the latter affords 120.5 gm (77%) of benzamidine hydrochloride, m.p. 161-163°C.
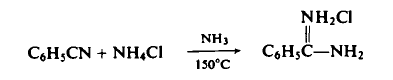 | | Biological Activity | Benzamidine hydrochloride is an reversible competitive inhibitor of trypsin-like serine proteases, with Kis of 97 μM, 21 μM, 20 μM and 110 μM for uPA, trypsin, tryptase and factor Xa, respectively. | | Safety Profile | Moderately toxic byintraperitoneal route. When heated to decomposition itemits toxic vapors of NOx, HCl, and Cl-. | | Synthesis | The general procedure for the synthesis of benzylcarbamidine hydrochloride from benzamidoxime was as follows: benzamidoxime (272 g, 2.0 mol, 1.0 eq.), methanol (500 mL), and Raney Ni catalyst (15 g) were added to a 1 L autoclave. A small amount of liquid ammonia was then added and the air in the kettle was displaced three times with nitrogen. Then hydrogen was introduced, the pressure in the kettle was maintained at 2-3 MPa, and the reaction temperature was controlled at 50°C. The reaction was continued until no more hydrogen was absorbed. Upon completion of the reaction, thermal filtration was performed and the filtrate was concentrated to dryness. The residue was cooled to 5 °C, a solid precipitate was precipitated and filtered to obtain gray crude benzylidine hydrochloride. Subsequently, the crude product was dissolved in ethanol (700 mL), heated until completely dissolved, and decolorized by adding activated charcoal and refluxing for 30 minutes. After performing thermal filtration, the filtrate was cooled to 0 °C, filtered to collect the white solid, and finally dried under vacuum at 50 °C to obtain 325 g of white solid benzylcarbamidine hydrochloride in 94.1% yield and 99.6% HPLC purity. | | in vitro | Benzamidine hydrochloride has weak inhibition for tPA and thrombin, with Kis of 750 μM and 320 μM, respectively. | | References | [1] F. MARKWARDT P. W H Landmann. Comparative Studies on the Inhibition of Trypsin, Plasmin, and Thrombin by Derivatives of Benzylamine and Benzamidine[J]. The FEBS journal, 1968, 6 4: 502-506. DOI:10.1111/j.1432-1033.1968.tb00473.x
[2] DEUTSCHER M P. Maintaining protein stability.[J]. Methods in enzymology, 2009, 463: 121-127. DOI:10.1016/s0076-6879(09)63010-x
[3] JAZWINSKI S M. Preparation of extracts from yeast.[J]. Methods in enzymology, 1990, 182: 154-174. DOI:10.1016/0076-6879(90)82015-t
[4] J W ENSINCK. Use of benzamidine as a proteolytic inhibitor in the radioimmunoassay of glucagon in plasma.[J]. Journal of Clinical Endocrinology & Metabolism, 1972, 35 3: 463-467. DOI:10.1210/jcem-35-3-463
[5] S L JEFFCOATE N W. Use of benzamidine to prevent the destruction of thyrotropin-releasing hormone (TRH) by blood.[J]. Journal of Clinical Endocrinology & Metabolism, 1974, 38 1: 155-157. DOI:10.1210/jcem-38-1-155 |
| | Benzamidine hydrochloride Preparation Products And Raw materials |
|