2-IODOPHENYLACETONITRILE manufacturers
- 2-IODOPHENYLACETONITRILE
-

- $6.60 / 1KG
-
2020-01-03
- CAS: 40400-15-5
- Min. Order: 1KG
- Purity: 97%-99%
- Supply Ability: 1kg -1000kg
|
| | 2-IODOPHENYLACETONITRILE Basic information |
| Product Name: | 2-IODOPHENYLACETONITRILE | | Synonyms: | O-IODOPHENYLACETONITRILE;2-IODOPHENYLACETONITRILE;2-IODOBENZYL CYANIDE;2-Iodophenylacetonitrile,96%;2-Iodobenzeneacetonitrile;2-(2-iodophenyl)acetonitrile;o-Iodobenzyl cyanide;2-Iodophenylacetonitrile 97% | | CAS: | 40400-15-5 | | MF: | C8H6IN | | MW: | 243.04 | | EINECS: | | | Product Categories: | | | Mol File: | 40400-15-5.mol | 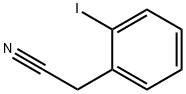 |
| | 2-IODOPHENYLACETONITRILE Chemical Properties |
| Boiling point | 113-120 °C0.5 mm Hg(lit.) | | density | 1.75 g/mL at 25 °C(lit.) | | refractive index | n20/D 1.618(lit.) | | Fp | >230 °F | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | Appearance | White to light yellow Solid | | Sensitive | Light Sensitive | | BRN | 2083106 | | InChI | 1S/C8H6IN/c9-8-4-2-1-3-7(8)5-6-10/h1-4H,5H2 | | InChIKey | FPSGTRJUQLYLHE-UHFFFAOYSA-N | | SMILES | Ic1ccccc1CC#N | | CAS DataBase Reference | 40400-15-5(CAS DataBase Reference) |
| Hazard Codes | Xn | | Risk Statements | 20/21/22-36/37/38 | | Safety Statements | 23-26-36 | | RIDADR | 3276 | | WGK Germany | 3 | | HazardClass | 6.1 | | PackingGroup | III | | Storage Class | 10 - Combustible liquids | | Hazard Classifications | Acute Tox. 4 Dermal
Acute Tox. 4 Inhalation
Acute Tox. 4 Oral
Eye Irrit. 2
Skin Irrit. 2
STOT SE 3 |
| | 2-IODOPHENYLACETONITRILE Usage And Synthesis |
| Uses | 2-Iodophenylacetonitrile may be used in the preparation of:
- 2-aminobiphen-2-ylacetonitrile
- ethyl (2-iodophenyl)iminoacetate hydrochloride
- 3,4-disubstituted 2-naphthalenamines
It may also be used in the preparation of the following nitriles:
- 2-(2-iodophenyl)-2-methylpropanenitrile
- 1-(2-iodophenyl)cyclopentanecarbonitrile
- 5-bromo-2-(2-iodophenyl)pentanenitrile
- 2-(2-iodophenyl)-2-propylpentanenitrile
- 1-(2-iodophenyl)cyclohexanecarbonitrile
- 1-(2-Iodophenyl)cyclopropanecarbonitrile
| | General Description | 2-Iodophenylacetonitrile is a 2-aryl substituted nitrile. It reacts with lactams to form ring-fused isoquinolinones via palladium-catalyzed carboxamidation in tandem with aldol condensation. | | Synthesis | Distilled dichloromethane (5 mL) was added to a 25 mL reaction tube under nitrogen protection. Subsequently, 161 mg (0.5 mmol) of iodobenzene diacetate was added. Next, 180 μL of trimethylsilyl trifluoromethanesulfonate (TMSOTf, 1.0 mmol) was added. The reaction mixture was stirred at room temperature for 5 min. After that, 198 mg (0.6 mmol) of 2-(tributylstannyl)acetonitrile was added at -78 °C and stirring was continued for 5 min, and the reaction process was monitored by thin layer chromatography (TLC). Upon completion of the reaction, 3 mL of saturated sodium bicarbonate solution was added to quench the reaction. The reaction was slowly warmed to room temperature and then extracted with dichloromethane (3 mL x 3). The organic phases were combined and dried over anhydrous sodium sulfate. It was subsequently concentrated under reduced pressure. Purification by column chromatography (Rf = 0.19, unfolding agent: petroleum ether/ethyl acetate = 40/1, v/v) afforded the target product 2-iodophenylacetonitrile as a white solid in 86% yield. | | References | [1] Angewandte Chemie - International Edition, 2018, vol. 57, # 29, p. 9078 - 9082
[2] Angew. Chem., 2018, vol. 130, # 29, p. 9216 - 9220,5
[3] Patent: CN108409602, 2018, A. Location in patent: Paragraph 0022; 0023; 0024 |
| | 2-IODOPHENYLACETONITRILE Preparation Products And Raw materials |
|