|
|
| | ETHYL PROPIONYL PYRUVATE Basic information |
| Product Name: | ETHYL PROPIONYL PYRUVATE | | Synonyms: | ETHYL PROPIONYL PYRUVATE;AKOS MSC-0253;Hexanoic acid, 2,4-dioxo-, ethyl ester;ethyl propionyl pyruvate,ethyl 2,4-dioxo hexanoate;2,4-Dioxo-hexanoic acid ethyl ester;2-Oxopentanoic propionic anhydride;2,4-dioxohexanoate;ETHYL PROPIONYL PYRUVATE USP/EP/BP | | CAS: | 13246-52-1 | | MF: | C8H12O4 | | MW: | 172.18 | | EINECS: | 236-228-4 | | Product Categories: | | | Mol File: | 13246-52-1.mol | 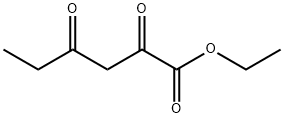 |
| | ETHYL PROPIONYL PYRUVATE Chemical Properties |
| Boiling point | 128-129 °C(Press: 11 Torr) | | density | 1.087g/ml | | FEMA | 3278 | ETHYL 2,4-DIOXOHEXANOATE | | storage temp. | 2-8°C | | pka | 6.75±0.46(Predicted) | | Appearance | Colorless to light yellow Liquid | | JECFA Number | 603 | | InChI | InChI=1S/C8H12O4/c1-3-6(9)5-7(10)8(11)12-4-2/h3-5H2,1-2H3 | | InChIKey | JGFBKJBAYISHAG-UHFFFAOYSA-N | | SMILES | C(OCC)(=O)C(=O)CC(=O)CC | | LogP | 0.35 | | EPA Substance Registry System | Ethyl 2,4-dioxohexanoate (13246-52-1) |
| | ETHYL PROPIONYL PYRUVATE Usage And Synthesis |
| Chemical Properties | Colorless liquid | | Definition | ChEBI: Ethyl 2,4-dioxohexanoate is a 3-acylpyruvic acid. | | Preparation | From methyl ethyl ketone and ethyl oxalate. | | Synthesis | 1. Under nitrogen protection, sodium ethoxide (10 g, 147 mmol) with anhydrous ethanol (65 mL) was added to a dry flask and heated to 70°C to dissolve completely. After cooling to 0 °C, a mixture of 2-butanone (13.16 mL, 147 mmol) and diethyl oxalate (19.96 mL, 147 mmol) was slowly added dropwise and subsequently warmed to room temperature. The reaction mixture was stirred for 1 hour and then heated to 80°C for 45 minutes. Cooled to room temperature and concentrated under reduced pressure to remove the solvent. The residue was dissolved in water (200 mL) and ethyl acetate (3 x 70 mL) to separate the organic phase. The aqueous phase was acidified to pH 2 with dilute sulfuric acid and extracted with ether (3×50 mL), the organic phases were combined, dried with anhydrous sodium sulfate, and concentrated under reduced pressure to give ethyl 2,4-dioxohexanoate as an amber colored oil 16.7 g (66% yield). Mass spectrum (CI) m/z: 173 (M+H)+.
2. Ethyl 2,4-dioxohexanoate (11.22 g, 65.15 mmol) was mixed with hydroxylamine hydrochloride (13.58 g, 195.5 mmol) in ethanol (200 mL) and heated to reflux for 1.5 hours. After cooling, the solvent was removed by concentration under reduced pressure. The residue was partitioned with water (150 mL) and ethyl acetate (2 x 150 mL), and the organic phases were combined, dried with anhydrous magnesium sulfate, and concentrated under reduced pressure to give an amber-colored oil (10.57 g). The crude product was purified by silica gel column chromatography (400 g silica gel, 20% ethyl acetate/hexane elution), the target fractions were combined, and concentrated under reduced pressure to afford ethyl 5-ethylisoxazole-3-carboxylate as a light-colored oil 8.95 g (81% yield). High resolution mass spectrometry (ESI) calculated value C8H11NO3+H: 170.0817, measured value 170.0824 (M+H)+.
3. An aqueous solution (35 mL) of sodium hydroxide (10.9 g, 273 mmol) was added to a solution of ethyl 5-ethylisoxazole-3-carboxylate (11.6 g, 68 mmol) in methanol (70 mL), and stirred at room temperature for 3 hours. The methanol was removed by concentration under reduced pressure, acidified with concentrated hydrochloric acid to pH 2. The acidic aqueous phase was extracted with dichloromethane (2 x 150 mL), then with 10% methanol/ dichloromethane (4 x 150 mL), the organic phases were combined, dried with anhydrous magnesium sulfate, and concentrated under reduced pressure to give 5.65 g of 5-ethylisoxazole-3-carboxylic acid as a white solid (58% yield). Mass spectrum (CI) m/z: 142 (M+H)+.
4. 5-Ethylisoxazole-3-carboxylic acid (1.41 g, 10 mmol) was dissolved in benzene (30 mL), oxalyl chloride (3.46 mL, 40 mmol) was added, and heated to reflux for 2 hours. After cooling, the solvent was removed by concentration under reduced pressure and the residue was azeotroped with dichloromethane to remove residual benzene. The resulting chloride was dissolved in acetone (15 mL), an aqueous solution of sodium azide (1.95 g, 30 mmol) was added (7 mL) and stirred vigorously for 1 hour. The acetone was removed by concentration under reduced pressure, and the residue was ground with water, filtered, washed with water and dried under vacuum to give 1.46 g of 5-ethylisoxazole-3-carbonyl azide as a white solid (88% yield).1H NMR (CDCl3,400MHz): δ1.34,2.85,6.46ppm.
5. 5-Ethylisoxazole-3-carbonyl azide (294 mg, 1.8 mmol) was mixed with 5-chloro-2,4-dimethoxyaniline (332 mg, 1.8 mmol) in anhydrous acetonitrile (20 mL) and heated to 70 °C for 20 h. The reaction was carried out at 70 °C for 20 min. After cooling, the solid was collected by filtration, washed with ether and dried under vacuum to give white solid Example 627 448 mg (78% yield). High resolution mass spectrometry (ESI) calculated value C14H16N3O4Cl+H: 326.0907, measured value 326.0909 (M+H)+. | | References | [1] Synthetic Communications, 2013, vol. 43, # 1, p. 110 - 117
[2] Journal of Agricultural and Food Chemistry, 2012, vol. 60, # 6, p. 1470 - 1479
[3] Chinese Chemical Letters, 2016, vol. 27, # 4, p. 566 - 570
[4] Patent: US2003/236287, 2003, A1. Location in patent: Page 47, 48
[5] Monatshefte fur Chemie, 2003, vol. 134, # 9, p. 1221 - 1227 |
| | ETHYL PROPIONYL PYRUVATE Preparation Products And Raw materials |
|