- HEXACHLORODISILANE
-
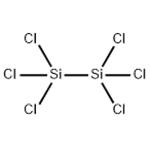
- $8.00 / 1KG
-
2025-09-25
- CAS:13465-77-5
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
- HEXACHLORODISILANE
-
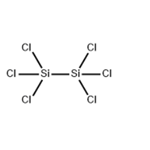
- $0.00 / 1KG
-
2025-04-04
- CAS:13465-77-5
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 1ton
- Hexachlorodisilane
-
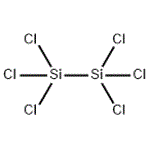
- $0.00 / 1kg
-
2023-06-19
- CAS:13465-77-5
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: kgs
|
| | Hexachlorodisilane Basic information |
| | Hexachlorodisilane Chemical Properties |
| Melting point | <0°C | | Boiling point | 144-145.5 °C (lit.) | | density | 1.562 g/mL at 25 °C (lit.) | | vapor pressure | 2.8hPa at 25℃ | | refractive index | n20/D 1.475(lit.) | | Fp | 78°C | | storage temp. | below 5° C | | solubility | sol CHCl3, CH2Cl2, benzene, THF; reacts violently,
producing toxic fumes, with H2O, alcohols. | | form | liquid | | Specific Gravity | 1.562 | | color | Colorless to Almost colorless | | Hydrolytic Sensitivity | 8: reacts rapidly with moisture, water, protic solvents | | Stability: | Stable, but reacts violently with water. Moisture sensitive. May be shock sensitive. Incompatible with water, moisture, acids, strong bases, oxidizing agents, alcohols. | | InChI | 1S/Cl6Si2/c1-7(2,3)8(4,5)6 | | InChIKey | LXEXBJXDGVGRAR-UHFFFAOYSA-N | | SMILES | Cl[Si](Cl)(Cl)[Si](Cl)(Cl)Cl | | CAS DataBase Reference | 13465-77-5(CAS DataBase Reference) | | EPA Substance Registry System | Disilane, hexachloro- (13465-77-5) |
| Hazard Codes | C | | Risk Statements | 14-34 | | Safety Statements | 26-36/37/39-45 | | RIDADR | UN 2987 8/PG 2 | | WGK Germany | 3 | | F | 3-10-21 | | TSCA | TSCA listed | | HazardClass | 8 | | PackingGroup | II | | HS Code | 28121049 | | Storage Class | 8A - Combustible corrosive hazardous materials | | Hazard Classifications | Skin Corr. 1B |
| | Hexachlorodisilane Usage And Synthesis |
| Chemical Properties |
Hexachlorodisilane is a colourless liquid, it is highly sensitive to moisture, and when exposed to air or water, it readily reacts, making it essential to handle the compound in a controlled, moisture-free environment.
| | Physical properties | bp 144–145.5 °C; d 1.562 g/cm3. | | Uses | Hexachlorodisilane can be used in deoxygenation and desulfurization of phosphine oxides, phosphine
sulfides, and amine oxides; reducing agent for nitro groups
and sulfur diimides. | | Preparation | Hexachlorodisilane is prepared in the chlorination of silicides such as e.g. calcium silicide. Idealized syntheses are as follows:
CaSi2 + 4 Cl2 → Si2Cl6 + CaCl2
| | Reactions | Hexachlorodisilane is a versatile chemical reagent that can be used in the following chemical reactions[1-2]:
(1) When hexachlorodisilane is heated with a catalytic amount of triphenylphosphine or triphenylarsine, it decomposes according to the equation nSi2Cl6 = nSiCl4 + (SiCl2)n. It does not react with trifluoromethylphosphine. Hexabromodisilane is also decomposed by trimethylamine.
(2) Hexachlorodisilane reacts with hydrogen chloride in the presence of pyridine, and the reaction formula is:
Si2Cl6 + 2HCl = 2SiCl4 + H2.
(3) Excess hydrogen bromide and hexachlorodisilane produce a mixture of hydrogen chloride and hydrogen bromide.
(4) Hexachlorodisilane reacts with methyl chloride in the presence of triphenylphosphine to produce a mixture of methyltrichlorosilane and silicon tetrachloride. When hexachlorodisilane and methyl bromide are heated together in the presence of triphenylphosphine, a mixture of methyl chloride and bromine is produced.
(5) Hexachlorodisiloxane reacts with sulfur trioxide to produce silicon dioxide, sulfur dioxide and hexachlorodisiloxane. | | General Description | Hexachlorodisilane (HCDS) is a chlorosilane used as a precursor for producing disilanes. It is a dioxidizer that is used in the production of silicon films and silicon nitride based films. | | Flammability and Explosibility | Non flammable | | Toxics Screening Level | The ITSL for hexachlorodisilane has been changed from 0.04 μg/m3 to 0.1 μg/m3 based on annual averaging time. | | References | [1] H.J. EMELéUS; Muhammad T. Reaction of hexachlorodisilane with bases and alkyl halides[J]. Journal of Inorganic and Nuclear Chemistry, 1967. DOI:10.1016/0022-1902(67)80468-8.
[2] B. S. SURESH D. P. Preparation of Hexafluorodisilane and Reactions of Hexafluorodisilane and Hexachlorodisilane with Sulfur Trioxide[J]. ACS Applied Electronic Materials, 1985. DOI:10.1246/BCSJ.58.1867.
|
| | Hexachlorodisilane Preparation Products And Raw materials |
|