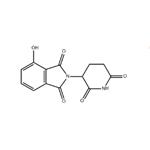
- 4-Hydroxy ThalidoMide
-
- $0.00 / 1kg
-
2023-03-15
- CAS:5054-59-1
- Min. Order: 1kg
- Purity: 98%Min
- Supply Ability: 1000kg
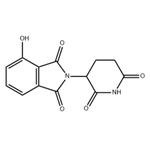
- 4-Hydroxy ThalidoMide
-
- $0.00 / 1KG
-
2022-09-24
- CAS:5054-59-1
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 1ton
|
| | 4-Hydroxy ThalidoMide Basic information |
| Product Name: | 4-Hydroxy ThalidoMide | | Synonyms: | 4-Hydroxy ThalidoMide;2-(2,6-Dioxo-piperidin-3-yl)-4-hydroxy-isoindole-1,3-dione;2-(2,6-dioxopiperidin-3-yl)-4-hydroxy-2,3-dihydro-1H-isoindole-1,3-dione;E3 ligase Ligand 2;Pomalidomide Impurity 15;Thalidomide Impurity 1;186942;1H-Isoindole-1,3(2H)-dione, 2-(2,6-dioxo-3-piperidinyl)-4-hydroxy- | | CAS: | 5054-59-1 | | MF: | C13H10N2O5 | | MW: | 274.23 | | EINECS: | | | Product Categories: | | | Mol File: | 5054-59-1.mol | 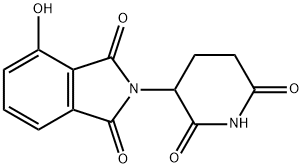 |
| | 4-Hydroxy ThalidoMide Chemical Properties |
| Melting point | 273-275 °C | | Boiling point | 568.3±45.0 °C(Predicted) | | density | 1.611±0.06 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | solubility | DMF: 12 mg/ml; DMSO: 10 mg/ml | | form | A solid | | pka | 6.82±0.20(Predicted) | | color | White to gray | | InChI | InChI=1S/C13H10N2O5/c16-8-3-1-2-6-10(8)13(20)15(12(6)19)7-4-5-9(17)14-11(7)18/h1-3,7,16H,4-5H2,(H,14,17,18) | | InChIKey | XMPJICVFSDYOEG-UHFFFAOYSA-N | | SMILES | C1(=O)C2=C(C(O)=CC=C2)C(=O)N1C1CCC(=O)NC1=O |
| | 4-Hydroxy ThalidoMide Usage And Synthesis |
| Description | 4-Hydroxy-thalidomide is the Thalidomide-based Cereblon ligand used in the recruitment of CRBN protein. It can be connected to the ligand for protein by a linker to form PROTACs. | | Uses | 2-(2,6-Dioxopiperidin-3-yl)-4-hydroxyisoindoline-1,3-dione is an intermediate for the synthesis of dTAG-13 (D710020), which is a degradation tag which was tested for its efficiency at depleting FKBP12F36V -MELK(sg3R) and was found to have significantly degraded FKBP12F36V -MELK(sg3R) within 4 hours.Used in the study of cancer research. | | Biochem/physiol Actions | Thalidomide hydroxylation can occur on glutarimide or phthalimide rings to yield metabolites of 4-hydroxy thalidomide. Interestingly, only metabolites phthaloyl glutamic acid and 4-hydroxy thalidomide have been tested for their anti-angiogenic activities. Hydroxylation of thalidomide on the C4-position of the phthalimide ring (4-hydroxy thalidomide) led to a weakly anti-angiogenic compound. 4-hydroxy thalidomide was not active in the CAM assay but was one of the most active compounds against HUVEC proliferation[1]. | | References | [1] M. G. Marks. “Effects of putative hydroxylated thalidomide metabolites on blood vessel density in the chorioallantoic membrane (CAM) assay and on tumor and endothelial cell proliferation.” Biological & pharmaceutical bulletin 356 1 (2002): 597–604. |
| | 4-Hydroxy ThalidoMide Preparation Products And Raw materials |
|