Chemical reaction and Synthesis of Chlorine Heptoxide
Feb 29,2024
Dichlorine heptoxide with the formula Cl2O7, it is a clear, oily, very volatile liquid. It reacts with water and organic compounds but dissolves in halocarbons.
Chemical reaction
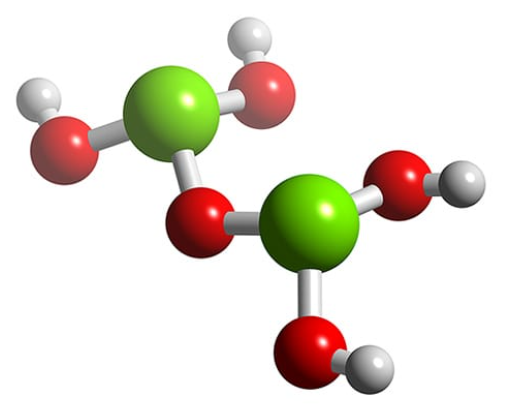
Dichlorine heptoxide reacts exothermically with water to yield perchloric acid.
Dichlorine heptoxide reacts with primary and secondary amines in carbon tetrachloride solution to yield perchloric amides:
2 RNH2 + Cl2O7 → 2 RNHClO3 + H2O
2 R2NH + Cl2O7 → 2 R2NClO3 + H2O
Unlike other chlorine oxides, dichlorine heptoxide does not attack sulfur, phosphorus, or paper at standard conditions.
Synthesis
This preparation is extremely dangerous because of the explosive and very oxidizing nature of Cl2O7.
Dichlorine heptoxide can be obtained by dehydrating perchloric acid with phosphorus pentoxide.
12 HClO4 + P4O10 → 4 H3PO4 + 6 Cl2O7
The reaction has to be performed under 0 °C, optimally between -70 °C to -10 °C. The resulting product has to be distilled very carefully under a vacuum to safely obtain pure dichlorine heptoxide.
Safety
Although it is the most stable chlorine oxide, Chlorine Heptoxide is a strong oxidizer as well as an explosive that can be set off with flame or mechanical shock, or by contact with iodine. Nevertheless, it is less strongly oxidizing than the other chlorine oxides and does not attack sulfur, phosphorus, or paper when cold.[1] It has the same effects on the human body as elemental chlorine and requires the same precautions.
- Related articles
- Related Qustion
Supplementation with pyridoxal 5'-phosphate monohydrate can synthesize neurotransmitters such as dopamine and serotonin, maintaining a healthy nervous system.....
Nov 4,2025Biochemical EngineeringChlorine Heptoxide
10294-48-1You may like