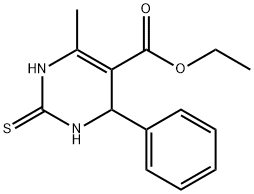
ETHYL 6-METHYL-4-PHENYL-2-THIOXO-1,2,3,4-TETRAHYDROPYRIMIDINE-5-CARBOXYLATE synthesis
- Product Name:ETHYL 6-METHYL-4-PHENYL-2-THIOXO-1,2,3,4-TETRAHYDROPYRIMIDINE-5-CARBOXYLATE
- CAS Number:33458-26-3
- Molecular formula:C14H16N2O2S
- Molecular Weight:276.35
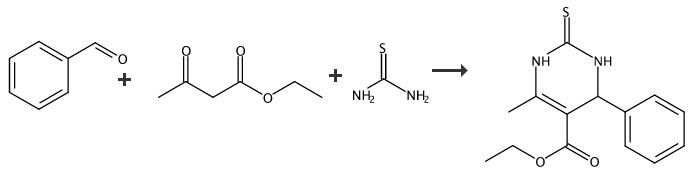
In a 10-mL round-bottom flask equipped with a condenser, a mixture of the aromatic aldehyde (1 mmol), ethyl acetoacetate (1 mmol), urea or thiourea (1.5 mmol) and [Dsbim]Cl (0.0376 g, 10 mol%) was stirred at 80 °C, and the resulting mixture was kept under continuous stirring for lengths of time as specified in Table 3, with the progress of the reaction was followed by TLC. After completion of the reaction, the mixture was cooled to room temperature, and 3 mL of water was added. The ionic liquid was dissolved in water and filtered for separation of the crude product. The separated product was washed twice with water (2 9 3 mL). For recycling the catalysts, after the solid products were thoroughly washed with water, the water containing the ionic liquid (IL is soluble in water) was evaporated under reduced pressure, and the ionic liquid was recovered and reused. The solid product was purified by a recrystallization procedure in ethanol. All of the desired product(s) were characterized by comparison of their physical data with those of known compounds. Entry: 13 Product: 4m; Yield: 90% Mp: 208-210°C; Lit. Mp: 205-206 [23]°C
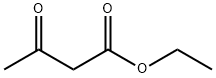
141-97-9
825 suppliers
$5.00/25g
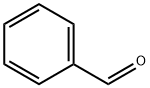
100-52-7
972 suppliers
$20.37/250g
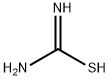
17356-08-0
2 suppliers
inquiry

33458-26-3
53 suppliers
$60.09/1gm:
Yield:33458-26-3 81%
Reaction Conditions:
with triethylamine in 1,4-dioxane;Reflux;
Steps:
General Procedure for the Synthesis of 7a-e
General procedure: A solution of the appropriate arylaldehyde 1a-e (0.01mol), ethyl acetoacetate (1.3 g, 0.01 mol) and thiourea(0.76 g, 0.01 mol) in 1,4-dioxan (40 mL) containingtriethylamine (1.0 mL) was heated under reflux for 6-8 h,then left to cool. The solid product formed upon pouringonto ice/water containing few drops of hydrochloric acidwas collected by filtration and crystallized from ethanol.
References:
Megally Abdo, Nadia Y. [Acta Chimica Slovenica,2015,vol. 62,# 1,p. 168 - 180]