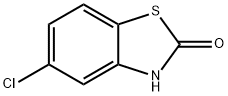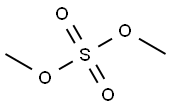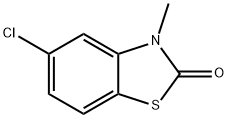
5-Chloro-3-methylbenzo[d]thiazol-2(3H)-one synthesis
- Product Name:5-Chloro-3-methylbenzo[d]thiazol-2(3H)-one
- CAS Number:2786-28-9
- Molecular formula:C8H6ClNOS
- Molecular Weight:199.66
Yield:2786-28-9 94%
Reaction Conditions:
with potassium carbonate in N,N-dimethyl-formamide at 20;
Steps:
31.i
Example 31; Preparation of 6-((butylthio)acetyJ)-5-c:h]oro-3-methyl-l,3-be?zothiazoi-2(3fl)-one (31); (i) 5-Chloro-3-methy-1 ,3-benzothiazol-2(3H)-one; 5-Chloro-2-benzothiazolor.e (5.010 g, 27.0 mmol) was dissolved in anhydrous DMF (60 ml) and anhydrous potassium carbonate (11.248 g, 81.4 mmol) and iodomethane (5.05 ml, 81.1 mmol) were added and the mixture stirred at room temperature overnight. The reaction mixture was poured into chloroform (240 ml) and filtered and the filtrate was evaporated to dryness. The resultant residue was dissolved in a mixture of ethyl aceLate (3(X) ml) and water (200 ml) then the phases were separated. The ethyl acetate phase was washed with water (2 x 100 ml) and brine (100 ml), dried over anhydrous magnesium sulfate, and filtered. The filtrate was evaporated to dryness to give the title compound (5.078 g, 94% yield) as a beige powder.η nmr (400 MHz, CDC13) δ 3.44 (s, 3H); 7.05 (d, /= 1.6Hz, I H); 7.16 (dd, J= 8.4, 2.0H7., lH); 7.34 (d, /= 8.4H7., I H).
References:
WO2007/70961,2007,A1 Location in patent:Page/Page column 83-84
3507-41-3
64 suppliers
$45.00/50mg
![5-Chloro-3-methylbenzo[d]thiazol-2(3H)-one](/CAS/20180703/GIF/2786-28-9.gif)
2786-28-9
8 suppliers
inquiry

2786-54-1
0 suppliers
inquiry
![5-Chloro-3-methylbenzo[d]thiazol-2(3H)-one](/CAS/20180703/GIF/2786-28-9.gif)
2786-28-9
8 suppliers
inquiry

29203-75-6
2 suppliers
inquiry
![5-Chloro-3-methylbenzo[d]thiazol-2(3H)-one](/CAS/20180703/GIF/2786-28-9.gif)
2786-28-9
8 suppliers
inquiry