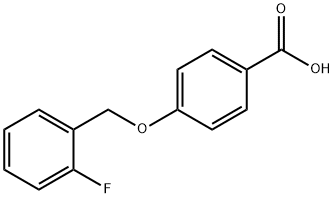
ASINEX-REAG BAS 13355090 synthesis
- Product Name:ASINEX-REAG BAS 13355090
- CAS Number:405-24-3
- Molecular formula:C14H11FO3
- Molecular Weight:246.23
![Benzoic acid, 4-[(2-fluorophenyl)methoxy]-, methyl ester](/CAS/20210305/GIF/128982-48-9.gif)
128982-48-9
1 suppliers
inquiry

405-24-3
24 suppliers
$49.00/200μmol
Yield:405-24-3 99%
Reaction Conditions:
Stage #1: methyl 4-(2-fluorobenzyloxy)benzoatewith water;potassium hydroxide in tetrahydrofuran;methanol at 20 - 25; for 36 h;
Stage #2: with hydrogenchloride in tetrahydrofuran;methanol;water;
Steps:
2
Methyl 4-(2-fluorobenzyloxy)benzoate (P1 , 10.0 g, 26.9 mmol) was dissolved in methanol (60 mL) and THF (90 mL). A 45 wt% potassium hydroxide solution (20 mL) was then added and the resulting exotherm was controlled by a water bath. After 1.5 days at 20-25 °C the solution became a thick suspension. Using a water bath to control the exotherm, 20 mL of concentrated HCI was added. The mixture was then concentrated to remove the THF and methanol and 150 mL water was added. The solid was isolated by filtration and washed with 50 mL water. After drying under vacuum, the title compound was isolated as a white crystalline solid (9.4 g, 99% yield).1H NMR (400 MHz, DMSO-d6) δ 7.95-7.89 (2H, m), 7.58 (2H, apparent td, J = 7.5, 1.7 Hz), 7.48-7.41 (1 H, m), 7.30-7.22 (2H, m), 7.16-7.10 (2H, m), 5.22 (2H, s).13C NMR (100 MHz, DMSO-d6) δ 167.3, 162.1 , 160.8 (d, J = 246 Hz), 131.7, 131.2 (d, J = 3.8 Hz), 131.0 (d, J = 8.3 Hz), 124.9 (d, J = 3.4 Hz), 123.8, 123.6, 115.8 (d, J = 21.0 Hz), 114.9, 64.2 (d, J = 3.4 Hz).LRMS (m/e) 247.2 [MH]+.
References:
WO2011/29762,2011,A1 Location in patent:Page/Page column 14-15
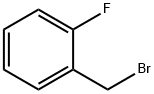
446-48-0
330 suppliers
$6.00/10g

405-24-3
24 suppliers
$49.00/200μmol
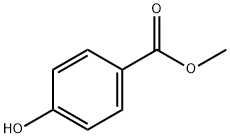
99-76-3
744 suppliers
$5.00/25g

405-24-3
24 suppliers
$49.00/200μmol
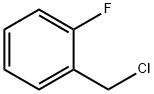
345-35-7
301 suppliers
$13.43/1gm:
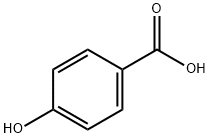
99-96-7
764 suppliers
$5.00/10g

405-24-3
24 suppliers
$49.00/200μmol