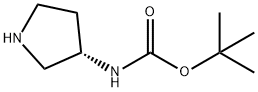
(S)-3-(Boc-amino)pyrrolidine synthesis
- Product Name:(S)-3-(Boc-amino)pyrrolidine
- CAS Number:122536-76-9
- Molecular formula:C9H18N2O2
- Molecular Weight:186.25
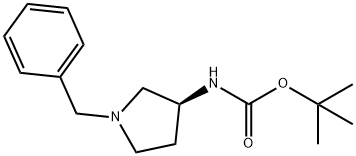
131852-53-4

122536-76-9
1. In a 500 mL four-necked flask fitted with a stirrer, thermometer, Dimroth condenser and titration funnel, 17.6 g (0.1 mol, optical purity 99.5% ee) of (S)-3-amino-1-benzylpyrrolidine was added. Subsequently, 158.7 g of water and 0.2 g of cationic DS (produced by Sanyo Chemical Industries, Ltd.) were added and the pH of the mixture was adjusted to 11 ± 0.5 using 48% aqueous sodium hydroxide solution. 2. The mixture was stirred at 50 to 60 °C while 26.2 g (0.12 mol) of di-tert-butyl dicarbonate (DiBoc) was added dropwise for about 2 hours. During the dropwise addition, the pH of the reaction solution was maintained at 11 ± 0.5 using 48% aqueous sodium hydroxide. 3. After completion of the dropwise addition, stirring was continued for 1 hour, after which the reaction solution was cooled to room temperature and the precipitated crystals were separated by filtration. 4. The resulting crystals were dried under vacuum at 50 °C to afford 26.0 g of (S)-1 -benzyl-3-tert-butoxycarbonylaminopyrrolidine in 94.1% yield, 99.1% chemical purity and 99.5% optical purity ee. 5. Using the same apparatus as in Step 1 , 26.0 g (optical purity 99.5% ee) of (S)-1 -benzyl-3-tert-butoxycarbonylaminopyrrolidine, 120 g of water, and 2.6 g of 5% Pd/C (PE-type, 55.27% water content, manufactured by NE Chemcat Corp.) were added. The reaction was stirred at 40°C reaction temperature for 10 hours under hydrogen atmosphere. 6. After confirming the completion of the reaction by GC analysis, the Pd/C was removed by filtration and the filtrate was concentrated to 30 g using an evaporator. 7. The concentrated product was mixed with toluene and concentrated again to 20 g to remove water by azeotropy. 8. 25 g of hexane was slowly added to the concentrated solution with stirring to precipitate crystals, followed by continued stirring in an ice bath for 2 hours. 9. The precipitated crystals were separated by filtration and dried under vacuum to afford 15.4 g of (S)-3-tert-butoxycarbonylaminopyrrolidine in 87.4% yield, 99.5% chemical purity, 99.5% optical purity ee, and 0.4% water content.

131852-53-4
72 suppliers
$20.00/5g

122536-76-9
294 suppliers
$7.00/1g
Yield:122536-76-9 87.4%
Reaction Conditions:
with hydrogen;5% palladium over charcoal in water at 40; for 10 h;
Steps:
3
A 500 ml four-neck flask equipped with a stirrer, a thermometer, a Dimroth condenser, and a titration funnel was loaded with (S)-3-amino-1-benzylpyrrolidine 17.6 g (0.1 mole, optical purity 99.5%/ee), water 158.7 g, and Cation DS (manufactured by Sanyo Chemical Industries, Ltd.) 0.2 g and the pH of the mixture was adjusted to be 11+/-0.5 with an aqueous 48% sodium hydroxide solution. While the mixture being stirred at 50 to 60°C, di-tert-butyl dicarbonate (hereinafter abbreviated as DiBoc) 26.2 g (0.12 mole) was dropwise added for about 2 hours. During the time, the reaction solution was adjusted at pH 11+/-0.5 with an aqueous 48% sodium hydroxide solution. After being stirred for further 1 hour, the reaction solution was cooled to a room temperature and precipitated crystal was separated by filtration. The crystal was vacuum dried at 50°C to obtain (S)-1-benzyl-3-tert-butoxycarbonylaminopyrrolidine 26.0 g. The yield was 94.1% and the chemical purity was 99.1 % and an optical purity was 99.5%ee. The same apparatus as that of Example 1 was loaded with (S)-1-benzyl-3-tert-butoxycarbonylaminopyrrolidine 26.0 g (optical purity 99.5%ee), water 120 g, and 5% Pd/C 2.6 g (PE type, 55.27% water content, manufactured by N. E. Chemcat Corp.) and the contents were stirred at reaction temperature of 40°C for 10 hours under hydrogen ventilation. When the reaction solution was analyzed by GC, and the peak of the raw material disappeared and other than toluene only 3-tert-butoxycarbonylaminopyrrolidine was detected. After completion of the reaction, Pd/C was removed by filtration and the filtrate was concentrated to 30 g by an evaporator. Next, the concentrated product was mixed with toluene and concentrated to 20 g to remove water by azeotropic boiling. While being mixed, the concentrated solution was mixed slowly with n-hexane 25 g to precipitate a crystal and further stirred for 2 hour in an ice bath. The precipitated crystal was separated by filtration and vacuum dried to obtain (S)-3-tert-butoxycarbonylaminopyrrolidine 15.4 g. The yield was 87.4%, the chemical purity was 99.5 area%, and an optical purity was 99.5 %ee. The water content was 0.4%.
References:
Toray Fine Chemicals Co., Ltd. EP1640364, 2006, A1 Location in patent:Page/Page column 10-11
122536-74-7
38 suppliers
inquiry

122536-76-9
294 suppliers
$7.00/1g
185057-49-2
13 suppliers
inquiry

122536-76-9
294 suppliers
$7.00/1g
![Carbamic acid, [1-(2-propenyl)-3-pyrrolidinyl]-, 1,1-dimethylethyl ester, (S)- (9CI)](/CAS/20200611/GIF/176970-11-9.gif)
176970-11-9
0 suppliers
inquiry

122536-76-9
294 suppliers
$7.00/1g
24424-99-5
869 suppliers
$13.50/25G

122536-76-9
294 suppliers
$7.00/1g