- Methyl trans-2-nonenoate
-

- $15.00 / 1KG
-
2021-08-12
- CAS:111-79-5
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
|
| | Methyl trans-2-nonenoate Basic information |
| Product Name: | Methyl trans-2-nonenoate | | Synonyms: | RARECHEM AL BF 0154;2-NONENOIC ACID METHYL ESTER;METHYL NONYLENATE;METHYL NONYLENOATE;METHYL NON-2-ENOATE;METHYL-2-NONENATE;FEMA 2725;Methyl trans-2-nonenoate | | CAS: | 111-79-5 | | MF: | C10H18O2 | | MW: | 170.25 | | EINECS: | 203-908-7 | | Product Categories: | | | Mol File: | 111-79-5.mol | 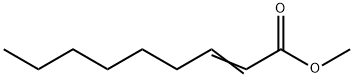 |
| | Methyl trans-2-nonenoate Chemical Properties |
| Hazard Codes | Xi,N | | Risk Statements | 36/38-51/53 | | Safety Statements | 26-61 | | RIDADR | UN 3082 9/PG 3 | | WGK Germany | 2 | | RTECS | RA9470000 | | toxicity | Both the acute oral LD50 value in rats and the acute dermal LD50 value in rabbits exceeded 5 g/kg (Moreno, 1975) |
| | Methyl trans-2-nonenoate Usage And Synthesis |
| Description | Methyl 2-nonenoate has a characteristic violet-like odor. May be
obtained in quantitative yields by treating methyl P-chlorocaproate
with sodium acetate, or by dehydrogenating the corresponding
saturated ester. | | Chemical Properties | Methyl 2-Nonenoate is a colorless to pale yellow liquid
with a green, floral note. It imparts freshness to floral fragrance compositions
and is recommended as a substitute for methyl 2-octynoate.The synthesis consists
of a condensation of heptanal withmalonic acid, decarboxylation, and subsequent
esterification with methanol. | | Physical properties | Methyl trans-2-nonenoate is colorless to slightly yellow liquid;strong violet-leaf odor. Stable; soluble in alco-hol. Combustible. | | Occurrence | Methyl trans-2-nonenoate has apparently not been reported to occur in nature. | | Uses | Perfumes. | | Preparation | Obtained in quantitative yields by treating methyl β-chlorocaproate with sodium acetate, or by dehydrogenating the cor�responding saturated ester. | | Definition | ChEBI: Methyl 2-nonenoate is a fatty acid ester. | | Taste threshold values | Taste characteristics at 5 ppm: green, waxy, oily, fruity, watermelon rind, pear- and apple-like. | | Synthesis Reference(s) | The Journal of Organic Chemistry, 54, p. 1831, 1989 DOI: 10.1021/jo00269a017
Synthetic Communications, 24, p. 2069, 1994 DOI: 10.1080/00397919408010217
Tetrahedron Letters, 30, p. 6555, 1989 DOI: 10.1016/S0040-4039(01)89020-1 | | Flammability and Explosibility | Not classified | | Trade name | Neofolione (Givaudan). | | Metabolism | 2-Nonenoic acid produced by the hydrolysis of methyl nonylenate will presumably pass through the normal pathways of fatty acid metabolism (Lehninger, 1970) |
| | Methyl trans-2-nonenoate Preparation Products And Raw materials |
|