- Cerium Ammonium Nitrate
-

- $10.00 / 10kg
-
2022-08-19
- CAS:16774-21-3
- Min. Order: 1kg
- Purity: 99.99%
- Supply Ability: 60 tons per month
- Ceric ammonium nitrate
-

- $1.00 / 1kg
-
2019-07-06
- CAS:16774-21-3
- Min. Order: 1kg
- Purity: 95%-99%
- Supply Ability: as request
|
| | Ceric ammonium nitrate Basic information |
| Product Name: | Ceric ammonium nitrate | | Synonyms: | Ammonium cerium(IV) nitrate puriss. p.a., ACS reagent, >=98.5% (RT);BARIUM HYDROXIDE SOLUTION PHE;TERRIFIC BROTH LIQUID;Ammonium cerium(IV) nitrate, 99.5% trace rare earth metals basis;Ammonium cerium(IV) nitrate, ACS, 98.5% min.;di-Ammonium Cerium(Ⅳ) Nitrate;AMMONIUM CERIUM(IV) NITRATE FOR ANALYSIS EMSURE ACS,REAG. PH EUR;Ammonium cerium(IV) nitrateACS reagent,
≥ 98.5% (Titration) | | CAS: | 16774-21-3 | | MF: | CeH4N7O18- | | MW: | 530.18 | | EINECS: | 240-827-6 | | Product Categories: | Inorganics;metal nitrate salts;Others;Oxidation;Synthetic Reagents;bc0001 | | Mol File: | 16774-21-3.mol | 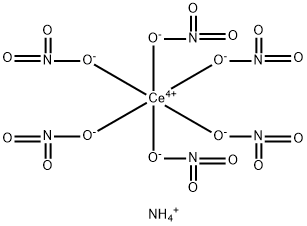 |
| | Ceric ammonium nitrate Chemical Properties |
| Melting point | 107-108 °C | | Boiling point | 83°C | | density | 1.10 g/mL at 20 °C | | vapor density | 2.3 (vs air) | | vapor pressure | 6.4 kPa (@ 20°C) | | storage temp. | Store at +5°C to +30°C. | | solubility | 919-1020g/l | | form | Powder/Solid | | color | Yellow to orange | | Specific Gravity | 1.045 | | PH | 0.61 (111.11g/l, H2O, 20℃) | | Odor | Slight characteristic odor | | Odor Threshold | 0.29 ppm | | Water Solubility | 1410 G/L (20 ºC) | | Merck | 14,1992 | | Stability: | Stable. Oxidizer - contact with combustible material may cause fire. Incompatible with strong reducing agents, combustible material. | | InChIKey | WIBGOERAEYJBOT-UHFFFAOYSA-N | | CAS DataBase Reference | 16774-21-3(CAS DataBase Reference) | | EPA Substance Registry System | Cerate(2-), hexakis(nitrato-.kappa.O)-, diammonium, (OC-6-11)- (16774-21-3) |
| Hazard Codes | Xi,O,Xn | | Risk Statements | 8-36/37/38-36/38-41-22 | | Safety Statements | 26-36-17-37/39 | | RIDADR | UN 1477 5.1/PG 2 | | WGK Germany | 3 | | F | 8 | | TSCA | Yes | | HS Code | 3822 00 00 | | HazardClass | 5.1 | | PackingGroup | III | | Toxicity | LD50 orally in Rabbit: > 300 - 2000 mg/kg |
| | Ceric ammonium nitrate Usage And Synthesis |
| Chemical Properties | yellow-orange crystals | | Physical properties | Reddish-orange monoclinic crystals; very soluble in water. | | Uses | A strongly oxidizing cerium preparation | | Uses | Used as oxidimetric standard. | | Uses | Ceric ammonium nitrate is used as a volumetric oxidizing reagent in many oxidation-reduction titrations. Cerium(IV) ion is a strong oxidant similar to permanganate ion. It is the most widely-used primary standard among all Ce(IV) compounds. Other applications of this compound are in organic oxidation reactions; and as a catalyst in polymerization of olefins. | | Preparation | Ceric ammonium nitrate is prepared by electrolytic oxidation of cerous nitrate in nitric acid to ceric nitrate, followed by the addition of ammonium nitrate solution. It is separated from the solution by crystallization. It may be prepared alternatively by dissolving cerium(II) oxide, CeO•H2O in concentrated nitric acid followed by treatment with ammonium nitrate. | | Reactions | The most important reactions of Ceric ammonium nitrate are the oxidations, attributed to Ce4+ ion in the solution. The standard reduction potential E° for the formal half-reaction: Ce4+ + e– Ce3+ in 1 M H2SO4 is 1.44 V. The oxidizing strength is comparable to permanganate (MnO4- ), bromate (BrO3-), and dichromate (Cr2O72-) anions. Analytical applications involve reactions with reductants such as sodium oxalate (Na2C2O4) or arsenic (III) oxide (As2O3) in the presence of iron, with ferroin (1,10–phenanthroline iron(II) complex) as the indicator. | | General Description | Ammonium cerium(IV) nitrate (Cerium(IV) ammonium nitrate, CAN) is a versatile reagent for oxidative electron transfer reactions. It participates in various novel carbon-carbon bond-forming reactions involved in one-pot synthesis of dihydrofurans, tetrahydrofurans and aminotetralins. Iodine/ammonium cerium(IV) nitrate has been employed in direct α-iodination of various ketones to afford the corresponding α-iodo ketones. CAN has been reported as an powerful one-electron oxidant. It also participates in various carbon-heteroatom bond-forming reactions. | | Hazard | Ceric ammonium nitrate is a powerful oxidizing agent. Precautions should be taken to avoid accidental contacts with orgnaic or other readily oxidizable substances. | | Flammability and Explosibility | Not classified | | Purification Methods | Ceric ammonium nitrate (125g) is warmed with 100mL of dilute HNO3 (1:3 v/v) and 40g of NH4NO3 until it dissolves, and filtered through a sintered-glass funnel. The solid which separates on cooling in ice is filtered off on a sintered funnel (at the pump) and air is sucked through the solid for 1-2 hours to remove most of the nitric acid. Finally, the solid is dried at 80-85o. |
| | Ceric ammonium nitrate Preparation Products And Raw materials |
|