- Simvastatin
-

- $0.00 / 1kg
-
2024-09-20
- CAS:79902-63-9
- Min. Order: 1kg
- Purity: 99%, Single impurity<0.1
- Supply Ability: 1 ton
- Simvastatin
-

- $0.00 / 1Kg/Bag
-
2024-09-18
- CAS:79902-63-9
- Min. Order: 1KG
- Purity: 98%-101%;USP
- Supply Ability: 500 KG
- Simvastatin
-

- $0.00 / 10g
-
2024-09-18
- CAS:79902-63-9
- Min. Order: 10g
- Purity: 98%
- Supply Ability: 50kg
Related articles - Simvastatin: uses and side effects
- Simvastatin is used to lower blood levels of "bad" cholesterol (low-density lipoprotein, or LDL), to increase levels of "good"....
- Sep 2,2024
|
| Product Name: | Simvastatin | | Synonyms: | (1s-(1-alpha,3-dro-4-hydroxy-6-oxo-2h-pyran-2-yl)ethyl)-1-naphthalenyleste;7-beta,8-beta(2s*,4s*),8a-beta))-alph;[1S-[1α,3α,7β,8β(2S^<*>^,4S^<*>^),8αβ]-2,2-Dimethylbutanoic acid 1,2,3,7,8,8α-hexahydro-3,7-dimethyl-8-[2-(tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-y1)ethyl]-1-naphthyl ester;Lodaiès;[(1S,3R,7R,8S,8aR)-8-[2-[(2R,4R)-4-hydroxy-6-oxo-oxan-2-yl]ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] 2,2-dimethylbutanoat;Butanoic acid, 2,2-dimethyl-, 1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-(2-(tetrahydro- 4-hydroxy-6-oxo-2H-pyran-2-yl)ethyl)-1-naphthalenyl ester, (1S-(1-alpha,3-alpha,7- beta,8-beta(2S*,4S*),8a-beta));[8-[2-(4-Hydroxy-6-oxo-tetrahydropyran-2-yl)ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl]2,2-dimethylbutanoate;2,2-Dimethylbutyric acid (1S,3R,7S,8S,8aR)-8-[2-[(2R,4R)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl]ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalene-1-yl ester | | CAS: | 79902-63-9 | | MF: | C25H38O5 | | MW: | 418.57 | | EINECS: | 616-751-8 | | Product Categories: | MOBIC;Chiral Reagents;Heterocycles;HMG-CoA reductase;API;Inhibitors;Intermediates & Fine Chemicals;Pharmaceuticals;API's;Simvastatin;APIs;Antibiotics | | Mol File: | 79902-63-9.mol | 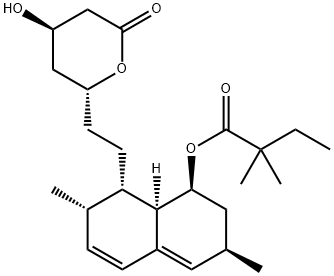 |
| | Simvastatin Chemical Properties |
| Melting point | 127-132 °C (lit.) | | alpha | D25 +292° (c = 0.5% in acetonitrile) | | Boiling point | 564.9±50.0 °C(Predicted) | | density | 1.11±0.1 g/cm3(Predicted) | | storage temp. | 2-8°C | | solubility | DMSO: ≥20mg/mL | | pka | 13.49±0.40(Predicted) | | form | solid | | color | white | | optical activity | [α]/D +275±25°, c = 1 in acetonitrile | | Merck | 14,8539 | | BCS Class | 2 | | Stability: | Stable for 2 years as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 2 months. | | CAS DataBase Reference | 79902-63-9(CAS DataBase Reference) | | NIST Chemistry Reference | Simvastatin(79902-63-9) | | EPA Substance Registry System | Butanoic acid, 2,2-dimethyl-, (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-[(2R,4R)-tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl]ethyl]-1-naphthalenyl ester (79902-63-9) |
| Provider | Language |
|
(1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-Hexahydro-3,7-dimethyl-8-[2-[(2R,4R)-tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl]ethyl]-1-naphthalenyl 2,2-dimethyl-butanoate
| English |
| | Simvastatin Usage And Synthesis |
| Lipid-lowering drugs | Simvastatin is a cholesterol-lowering drug of statin, is a synthetic derivative of Aspergillus terreus fermentation product, is used to control blood cholesterol levels and prevent cardiovascular disease.
Simvastatin belongs to methyl hydroxyl coenzyme A (HMG-COA) reductase inhibitors, inhibits synthesis of endogenous cholesterol, and is the lipid regulator. Has a role in reducing cholesterol (TC) content of serum, liver and aorta in hyperlipidemia rabbits reducing levels of very low density lipoprotein cholesterol (VLDL-C) and low-density lipoprotein cholesterol (LDL-C). | | Indications | 1. Hyperlipidemia:
(1) For patients with primary hypercholesterolemia, heterozygous familial hypercholesterolemia or mixed hypercholesterolemia, when diet and other non-drug treatment is not ideal, Simvastatin can be used to reduce the increased total cholesterol, LDL cholesterol, apolipoprotein B and triglycerides. And Simvastatin increases high-density lipoprotein cholesterol, thereby reducing the LDL/HDL and total cholesterol/HDL ratio.
(2) For patients with homozygous familial hypercholesterolemia, when diet and non-diet is not ideal, Simvastatin can be used to reduce elevated total cholesterol, LDL cholesterol and apolipoprotein B.
2. Coronary heart disease:
Coronary heart disease, Simvastatin can be used to:
(1) reduce the risk of death.
(2) reduce risk of coronary heart disease death and nonfatal myocardial infarction.
(3) reduce risk of stroke and transient ischemic.
(4) reduce risk of myocardial revascularization (coronary artery bypass grafting and percutaneous coronary balloon angioplasty).
(5) delay the progression of atherosclerosis in the arteries, including the all-new lesions and the occurrence of full clogging.
The above information is edited by the chemicalbook of Liu Yujie. | | Chemical properties | White crystalline powder, odorless. Soluble in ethanol, acetone or acetonitrile, difficult soluble in ether, practically insoluble in water. Melting point is 135~138 ℃. | | Uses | 1. HMG-CoA reductase inhibitors, can reduce the concentration of serum total cholesterol level, inhibit synthesis of cholesterol. For treatment of primary hypercholesterolemia with cholesterol levels greater than 7.8mmol/L which is invalid or not tolerated in other treatments.
2. Has role in reducing cholesterol, low-density lipoprotein cholesterol and very low density lipoprotein cholesterol. | | Production methods | Lovastatin (I, 50.30 g, 0.124 mol) and n-butylamine (42 ml, 0.42 mo1), at 25 ℃: mixed, then heated at 80 ℃ for 1h. Cooled at 25 ℃, evaporated excess butylamine under reduced pressure to give 59.4g compound (Ⅱ), yield was 100%, wass used directly for the next step.
Crude product of above obtained compound (Ⅱ), is dissolved in 132 ml of dimethylformamide at 25 ℃, added imidazole (19.59 g, 0.288 mol) and tert-butyldimethylsilyl chloride (TBSCl, 44.4 g, 0.288 mol) , heated at 60 ℃ for 60 h. Cooled to 12 ℃, added anhydrous methanol (5.8 ml, 0.143 mol), stirred at 15 ℃ for 30min, added 1.5 L cyclohexane and 750 ml 5% sodium bicarbonate solution and stirred vigorously. Stratified, cyclohexane layer was separated, washed with 750ml 5% sodium bicarbonate solution and 750ml water. Concentrated to 580ml at atmospheric pressure, then add 600ml of tetrahydrofuran dried by molecular sieve, and then concentrated to 870 ml under atmosphere pressure. 86.9 g compound (Ⅲ) showed in HPLC, yield was 99%, was used directly for the next reaction.
Pyrrolidine (25.1ml, 0.301 mol)dried by molecular sieve was mixed with 192 ml of tetrahydrofuran, cooled to-18 ℃, added n-butyl lithium in hexane solution (1.60 mol/L, 181 ml, 0.290 mol), maintained at-10 ℃ about 15min addition was completed, then further reacted at-20 ℃ for 15 min. Thus obtained pyrrolidinyl lithium solution, cooled to-20 ℃ for use. The etrahydrofuran-cyclohexane solution of above obtained compound (Ⅲ) was cooled to-35 ℃, under vigorous stirring, added pyrrolidinyl lithium solution cooled to-20 ℃, maintained at-30 ℃. Addition was completed, stirred at-35 ~-30 ℃ for 2h, then added iodomethane all at once (12.2 ml, 0.196 mol), due to the exothermic reaction at this time, the temperature will rise to 20 ℃, then cooled to-30 ℃, and stirred at this temperature for 1 h. Rose to-10 ℃, stirred for 20 min. added 550ml of water, stirred vigorously for 10min, the organic layer was separated, washed with 550 ml 10 ℃ of 1 mol/L hydrochloric acid. Concentrated to 20% volume under reduced pressure, which contained the compound (Ⅳ), was used directly for the next reaction.
Added 690ml of methanol into the above obtained compound (Ⅳ), added 45.7 ml of water and methanesulfonic acid (1.5 ml, 0.023 mol), stirred at 30 ℃ for 5 h, i.e. formed compound ( V), was used directly for the next step reaction.
For a solution of above obtained compound (V) , at 25 ℃ 373 ml 2 mol/L sodium hydroxide solution was added, heated at atmospheric pressure, and subjected to distillation, when the temperature of gas phase reached 72~73 ℃, the liquid temperature reached 78~80 ℃, no longer collected distilled distillate. The remaining solution was stirred at reflux for 2h, then cooled to 40 ℃. Most of the methanol was distilled off under reduced pressure, then add 228ml water to dilute. Cooled to 10 ℃, adjusted with 3 mol L/HCl PH = 6~7. And 990 ml of ethyl acetate was added, continued to adjust PH = 5.0 with 3mol/L hydrochloric acid. After stirred to stratify, the ethyl acetate layer was separated, 225 ml of methanol was added. In 1 h, added 75 ml ammonia-methanol: solution (1:3), and then stirred at 45 ℃ for 15 min. In 2.5 h, slowly cooled to-10 ℃, and at-10 ℃ stirred for 1~2 h. Filtered ammonia salts (Ⅵ) crystals, washed with cold ethyl acetate-methanol (3.5:1), at 35 ℃ dried overnight to give 51.37 g ammonia salts (Ⅵ), a yield was 90.9% (calculated by lovastatin). Recrystallized with acetonitrile to obtain a sample for analysis.
The crude product of ammonium salt (Ⅵ) was suspended in 1.03 L toluene, inlet nitrogen, heated at 100 ℃ for 6 h. Cooled to 25 ℃, added 2.5 g Darco KB, stirred at 25 ℃ for 30 min. Filtered and the filtrate was concentrated to grease under reduced pressure, added 140 ml of cyclohexane and concentrated. Then add 600ml of cyclohexane, reflux and completely dissolved. Cooled to 10 ℃, stirred for 1 h. Filtered and collected crystals, washed with 25 0ml of cold cyclohexane, dried at 30~35 ℃ under vacuum to give 44.6 g Simvastatin, yield was 94.2%. Recrystallized with methanol-water to give a sample for analysis. | | Category | Toxic substances. | | Toxicity grading | Poisoning | | Acute toxicity | Oral-rat LD50: 4438 mg/kg; Oral-mouse LD50: 3000 mg /kg. | | Flammability hazard characteristics | Combustible; fire releases spicy smoke. | | Storage characteristics | Treasury is low temperature, ventilation, dry; in dark. | | Extinguishing agent | Water, carbon dioxide, dry, sandy soil. | | Description | Simvastatin is an once-daily hypolipemic, an analog of lovastatin indicated for the
treatment of atherosclerosis. In patients with Type IN or IIB hypercholesterolemia,
simvastatin reportedly produces significant reductions in total serum cholesterol, LDL,
mglycerides and apolipoprotein-B, while HDL and apolipoprotein-A levels are increased. | | Chemical Properties | White Powder | | Originator | Merck (USA) | | Uses | A HMGCR inhibitor and anti-proliferative agent. | | Uses | anti-hyperlipoproteinemic, 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor | | Uses | antiinflammatory | | Uses | Simvastatin is semi-synthetic, slightly more hydrophobic, analogue of lovastatin. Like lovastatin, simvastatin is a specific inhibitor of HMG-CoA reductase and is used therapeutically to reduce LDL cholesterol. More recently, the statins have become important biochemical probes in cell biology. Their involvement in many events can be correlated to their primary mode of action, however, the mechanism of action of many other effects is less apparent. | | Uses | Simvastatin is a synthetic derivative of a fermentation product of Aspergillus terreus. A competitive inhibitor of HMG-CoA reductase. A synthetic analog of Lovastatin. Antilipemic. Simvastatin, the dr
ug, is sold under the trade name Zocor. | | Uses | Simvastatin is a synthetic derivate of a fermentation product of Aspergillus terreus. A competitive inhibitor of HMG-CoA reductase. A synthetic analog of Lovastatin. Antilipemic. Simvastatin, the drug, is sold under the trade name Zocor. | | Definition | ChEBI: A carbobicyclic compound that is lovastatin in which the 2-methylbutyrate ester moiety has been replaced by a 2,2-dimethylbutyrate ester group. It is used as a cholesterol-lowering and anti-cardiovascular disease drug. | | Manufacturing Process | A suspension of Lovastatin (350 g, 0.865 mmol), phenylboronic acid (110.8 g,
0.909 mmol) and toluene (1.75 L) was heated under a nitrogen atmosphere
at 100-105°C for 55 min. The water was separated from the reaction mixture.
The solution was cooled and 1.39 L of toluene was removed by vacuum
distillation at 40-50°C. The concentrated solution was treated with hexanes
(3.15 L) at 40-50°C. The resulting suspension was cooled to 0-5C for 2 hours
and the product was filtered and washed with hexanes (350 mL). The product
was dried at 35-40°C under vacuum to provide 427.9 g (37%) of lovastatin
phenylboronate at >99% purity by HPLC.
A 2 L 3-necked flask was charged with pyrrolidine (56 mL, 0.67 mol) and dry
THF (453 g) under a nitrogen atmosphere. n-Butyl lithium (419 mL, 1.6 M
hexane solution, 0.67 mol) was added dropwise at -20°C over a period of 1
hour. The solution was maintained at this temperature for 30 min and then
cooled to -55°C. A solution of lovastatin phenylboronate (101.7 g, 0.20 mol)
in 274.7 g of THF was cooled to -50°C and then added to the cold lithium
pyrrolidide solution at a rate such that the internal temperature was between
-50°-55°C during the addition. The mixture was held at this temperature for 4
hours and then methyl iodide (116.4 g, 0.82 mol) was added at a
temperature below -55°C. The reaction was stirred for 13 hours at -20°C and
then quenched with 500 mL of 2 M HCl at a temperature below 0°C. After
warming to 20°C, the layers were separated and the aqueous layer was
extracted with ethyl acetate. The combined organic layers were washed with
5% NaHSO3 solution and deionized water. The solution was filtered through a
Celite pad and concentrated to yield 102.8 g (98.4%) of crude Simvastatin
phenylboronate at >95% purity by HPLC. A portion of the above material
(50.0 g) was charged into a nitrogen purged flask with acetonitrile (100 mL).
The suspension was heated at 110°C for 3 hours and then cooled to - 10°C for 1 hour. The product was filtered and washed with 25 mL of acetonitrile and
dried under vacuum to provide 43.7 g of Simvastatin phenylboronate at >99%
purity by HPLC.
A suspension of simvastatin phenylboronate (30.0 g) and 1,3-propanediol
(450 mL) was heated at 105-107°C at 0.2 mm Hg. After 1 hour, 182 mL of
distillate was collected and the reaction was cooled to 20-25°C. Deionized
water (270 mL) was added and toluene (3 times 75 mL) was used to extract
the mixture. The combined toluene layers were washed with water (60 mL).
The organic solution was heated at reflux for 1 hour and water was
azeotropically removed. The solution was concentrated to a final volume of 24
mL under vacuum at 48-50°C. To the concentrated solution was added
hexanes (215 mL) over 10 min. The resulting slurry was cooled to 0-5°C and
filtered. The crude Simvastatin was washed at 0-5°C with hexanes and dried
under vacuum to yield 21.0 g (88%) of Simvastatin. | | Brand name | Zocor (Merck);Zocord. | | Therapeutic Function | Antihyperlipidemic | | General Description | Simvastatin, 2,2-dimethyl butanoic acid,1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-(tetrahydro-4-hydroxy-6-oxo-2-pyran-2-yl)ethyl]-1-naphthalenyl ester(Zocor), is an analog of lovastatin. These two drugs havemany similar properties. Both drugs, in the prodrug form,reach the liver unchanged after oral administration, wherethey undergo extensive metabolism to several open-ring hydroxyacids, including the active -hydroxy acids. They arealso highly bound to plasma proteins. These actions makethe bioavailability of simvastatin rather poor but better thanthat of lovastatin, which has been estimated to be 5%. | | Biological Activity | HMG-CoA reductase inhibitor; decreases levels of low density lipoprotein. Has multiple biological effects including bone formation stimulation, inhibition of smooth muscle cell proliferation and migration, and anticancer and anti-inflammatory activity. | | Biochem/physiol Actions | Simvastatin is a specific inhibitor of HMG-CoA reductase, the enzyme that catalyzes the conversion of HMG-CoA to mevalonate, an early step in cholesterol biosynthesis. It is used in the treatment of hypercholesterolemia, as it reduces levels of low-density lipoproteins and triglycerides, and raises high-density lipoprotein levels. Simvastatin is a lactone that is readily hydrolyzed in vivo to the corresponding β-hydroxyacid, and can be activated prior to use with NaOH in EtOH treatment. It is a synthetic analog of lovastatin (Cat. No. M2147). | | Clinical Use | HMG CoA reductase inhibitor:
Primary hypercholesterolaemia | | target | TNF-α | NF-kB | MMP(e.g.TIMP) | p65 | JNK | Beta Amyloid | TGF-β/Smad | NO | NOS | STAT | IFN-γ | | Drug interactions | Potentially hazardous interactions with other drugs
Anti-arrhythmics: increased risk of myopathy with
amiodarone - do not exceed 20 mg of simvastatin1
;
increased risk of myopathy with dronedarone.
Antibacterials: increased risk of myopathy with
clarithromycin, daptomycin, erythromycin and
fusidic acid - avoid; possibly increased myopathy
with azithromycin; concentration possibly reduced
by rifampicin.
Anticoagulants: effects of coumarins enhanced.
Antiepileptics: concentration reduced by
carbamazepine and eslicarbazepine.
Antifungals: increased risk of myopathy
with fluconazole, itraconazole, posaconazole,
ketoconazole, voriconazole and possibly miconazole
- avoid; possibly increased risk of myopathy with
imidazoles.
Antivirals: increased risk of myopathy with
atazanavir, indinavir, lopinavir, ritonavir or saquinavir
and possibly fosamprenavir, lopinavir or tipranavir -
avoid; concentration reduced by efavirenz; avoid with
boceprevir, dasabuvir, ombitasvir, paritaprevir and
telaprevir; possible increased risk of myopathy with
ledipasvir - reduce simvastatin dose; concentration
increased by simeprevir - consider reducing
simvastatin dose.
Calcium-channel blockers: increased risk of
myopathy with verapamil, diltiazem and amlodipine
- do not exceed 20 mg of simvastatin.1
Ciclosporin: increased risk of myopathy - avoid.1
Cobicistat: avoid with simvastatin.
Colchicine: possible increased risk of myopathy.
Grapefruit: increased risk of myopathy - avoid.
Hormone antagonists: possibly increased risk of
myopathy with danazol - avoid.1
Lipid-lowering agents: increased risk of myopathy
with fibrates - do not exceed 10 mg of simvastatin
except with fenofibrate1
; gemfibrozil - avoid;
concentration increased by lomitapide - do not
exceed 40 mg of simvastatin; increased risk of
myopathy with nicotinic acid.
Ranolazine: concentration increased by ranolazine,
maximum dose of simvastatin is 20 mg.
Ticagrelor: concentration of simvastatin increased;
maximum dose of simvastatin is 40 mg | | Metabolism | Simvastatin is absorbed from the gastrointestinal tract
and must be hydrolysed to its active β-hydroxyacid form.
Other active metabolites have been detected and several
inactive metabolites are also formed. Simvastatin is a
substrate for the cytochrome P450 isoenzyme CYP3A4
and undergoes extensive first-pass metabolism in the
liver, its primary site of action. Less than 5% of an oral
dose has been reported to reach the circulation as active
metabolites.
Simvastatin is mainly excreted in the faeces via the bile
as metabolites. About 10-15% is recovered in the urine,
mainly in inactive forms. | | storage | Desiccate at -20°C | | References | References/Citations
1) Merck 14:8539
2) Hancock et al. (1989), All ras proteins are polyisoprenylated but only some are palmitoylated; Cell, 57 1167
3) Ose et al. (2000), Lipid-altering efficacy and safety of simvastatin 80 mg/day: long-term experience in a large group of patients with hypercholesterolemia. World Wide Expanded Dose Simvastatin Study Group Clin. Cardiol. 23 39
4) Matsuzaka et al. (2016) Characterization and functional analysis of extracellular vesicles and muscle-abundant miRNA in C2C12 myocytes and Mdx mice; PLoS One 11(12) e0167811 [Focus Biomolecules Citation] |
| | Simvastatin Preparation Products And Raw materials |
|