Lidocaine Hcl: Uses, Side effects, Overdose, Interactions
Sep 16,2021
Chemistry
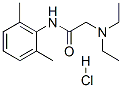
C14H12N2O.HCl
2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide
Brand name Xylocaine
General
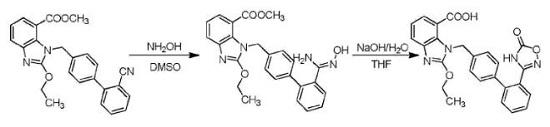
Lidocaine hydrochloride was synthesized by Löfgren and Lundquist in 1943, and was clinically introduced in 1948. It remains one of the most widely used local anaesthetics. It can be administered parenterally for a peripheral nerve block (PNB), intravenously, or applied topically at strengths of 2–4%. The addition of epinephrine 1:200 000 to 1:100 000 slows the vascular absorption of lidocaine and prolongs its effects
Available concentrations
0.5%
1% with/without epinephrine 1/200 000
2% with/without epinephrine 1/200 000
4% (topical)
5% (medicated plaster)
Dose mg/kg
3 (without epinephrine)
7 (with epinephrine)
Additional dose guidelines
600 mg by topical application in 24 h
200 mg for PNB
500 mg for PNB (with epinephrine)
Elderly: doses may need to be reduced depending on age and physical state
Onset time: Fast (2–5 min)
Duration of anaesthesia: Short (45–60 min)
Peak plasma levels: 15–30 min
Relative toxicity:Low: 1 (toxicity values for the other drugs will be compared to lidocaine)
Mean half life:1.5 hr
Protein binding: 65–75%
Volume of distribution: 90 L
Metabolism
Lidocaine is metabolized by the liver with only a small amount (3%) found
unchanged in urine. The three main mechanisms of metabolism are shown below:
N-de-ethylation > monoethylglycinexylidide (MEGX) > glycinexylidide
Hydrolysis of glycinexylidide
5-hydroxylation of lidocaine’s benzene ring
Lidocaine possesses convulsant activity. Hepatic disease or reduced hepatic blood
flow (as in congestive cardiac failure) will lower metabolic capacity
Potency: Medium
Excretion:Renal
Relative lipid solubility: 150
pKa: 7.86
Drug interactions
Cimetidine – inhibits hepatic metabolism
Propranolol – reduces systemic clearance by up to 40% by reducing hepatic metabolism
Additional information
Lidocaine is contraindicated in patients with known hypersensitivity to local
anaesthetics of the amide type and in patients with porphyria. Reactions due to
overdose with lidocaine (high plasma levels) are systemic and involve the central
nervous and cardiovascular systems. Effects include medullary depression, tonic and
clonic convulsions, and cardiovascular collapse.
Solutions in multidose vials may contain hydrobenzoate derivatives and have been
associated with allergic reactions in some patients. As with all of the amide local
anaesthetics protein binding is reduced in the neonate (50% versus 64% in the
adult), which necessitates reduced doses if adverse reactions are to be avoided.
- Related articles
- Related Qustion
- Lidocaine hydrochloride for Chronic Pain Treatment Jan 25, 2024
Lidocaine hydrochloride effectively reduces non-specific chronic pain, neuropathic pain, musculoskeletal pain and improves quality of life. Further studies needed for musculoskeletal pain.
- Lidocaine hydrochloride: mechanism of action, pharmacokinetics and activities Jun 28, 2023
Lidocaine hydrochloride is a medication that works by blocking sodium channels in neuronal cell membranes, preventing the initiation and transmission of nerve impulses.
- What is Lidocaine hydrochloride? Oct 10, 2020
Lidocaine hydrochloride is a local anesthetic and antiarrhythmic drug. It is clinically used for infiltration anesthesia, epidural anesthesia, surface anesthesia and nerve conduction block.
2-Nitro-5-chloroaniline is an important pharmaceutical and veterinary drug intermediate. It has been applied in the upgraded product of veterinary drug albendazole-fenbendazole, and has a broad market prospect.....
Sep 15,2021APIAzilsartan (Azilsartan), the trade name is Azilva,It was approved to be marketed in Japan on January 18, 2012. It has now been approved for marketing in the United States and the European Union.....
Sep 16,2021APILidocaine hydrochloride
73-78-9You may like
Lidocaine hydrochloride manufacturers
- Lidocaine hydrochloride
-
- $31.00 / 5g
- 2025-08-20
- CAS:73-78-9
- Min. Order: 5g
- Purity: 0.99
- Supply Ability: 25kg
- Lidocaine hydrochloride
-

- $10.00 / 1kg
- 2025-08-20
- CAS:73-78-9
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 10000
- Lidocaine hydrochloride
-

- $35.00 / 1kg
- 2025-08-20
- CAS:73-78-9
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 300tons