| Identification | More | [Name]
1,4-Butanediol vinyl ether | [CAS]
17832-28-9 | [Synonyms]
1,4-BUTANEDIOLMONOVINYL ETHER
1,4-BUTANEDIOL VINYL ETHER
4-HYDROXYBUTYL VINYL ETHER
4-VINYLOXYBUTANOL
BUTANEDIOL MONOVINYL ETHER
HYDROXYBUTYL VINYL ETHER
TETRAMETHYLENE GLYCOL MONOVINYL ETHER
VINYL 4-HYDROXYBUTYL ETHER
4-(ethenyloxy)-1-butano
4-(ethenyloxy)-1-Butanol
4-vinyloxy-butan-1-ol
Stabilizedwithpotassiumhydroxide
4-Hydroxybutyl vinyl ether, stabilized, 99%
1-Butanol, 4-(ethenyloxy)-
4-(ETHENYLOXY)BUTAN-1-OL
1,4-BUTANEDIOL VINYL ETHER 99%
Tetramethylene Glycol Monovinyl Ether (stabilized with KOH)
HBVE
4-Hydroxybutyl vinyl ether, 99%, stabilized
4-(Ethenyloxy)butane-1-ol | [EINECS(EC#)]
241-793-5 | [Molecular Formula]
C6H12O2 | [MDL Number]
MFCD00080697 | [Molecular Weight]
116.16 | [MOL File]
17832-28-9.mol |
| Chemical Properties | Back Directory | [Appearance]
colorless to yellow liquid | [Melting point ]
-33 °C
| [Boiling point ]
95 °C/20 mmHg (lit.) | [density ]
0.939 g/mL at 25 °C(lit.)
| [vapor pressure ]
0.3 hPa (20 °C) | [refractive index ]
n20/D 1.444(lit.)
| [Fp ]
185 °F
| [storage temp. ]
Store at <= 20°C. | [solubility ]
75g/l | [form ]
clear liquid | [pka]
15.04±0.10(Predicted) | [color ]
Colorless to Light yellow | [Specific Gravity]
0.93 | [explosive limit]
1.4-9.9%(V) | [Water Solubility ]
75 g/L (20 ºC) | [InChI]
1S/C6H12O2/c1-2-8-6-4-3-5-7/h2,7H,1,3-6H2 | [InChIKey]
HMBNQNDUEFFFNZ-UHFFFAOYSA-N | [SMILES]
OCCCCOC=C | [LogP]
0.43 at 25℃ | [CAS DataBase Reference]
17832-28-9(CAS DataBase Reference) | [EPA Substance Registry System]
17832-28-9(EPA Substance) |
| Questions And Answer | Back Directory | [Uses]
4-Hydroxybutylvinyl ether is a commercially available polymer chemical, polymer material, and organic synthesis intermediate. It can undergo self-polymerization under the action of sulfonic acid. In synthetic transformation, its hydroxyl groups can be converted into aldehyde groups under Swern oxidation conditions. |
| Safety Data | Back Directory | [Symbol(GHS) ]

GHS07 | [Signal word ]
Warning | [Hazard statements ]
H302-H319-H412 | [Precautionary statements ]
P264-P270-P273-P280-P301+P312-P305+P351+P338 | [Hazard Codes ]
Xn | [Risk Statements ]
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed .
R36/37/38:Irritating to eyes, respiratory system and skin .
R22:Harmful if swallowed. | [Safety Statements ]
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S36:Wear suitable protective clothing .
S24/25:Avoid contact with skin and eyes . | [RIDADR ]
NA 1993 / PGIII | [WGK Germany ]
1
| [Autoignition Temperature]
225 °C DIN 51794 | [TSCA ]
TSCA listed | [REACH Registrations]
Active | [HS Code ]
29091990 | [Storage Class]
10 - Combustible liquids | [Hazard Classifications]
Acute Tox. 4 Oral
Aquatic Chronic 3
Eye Irrit. 2 | [Toxicity]
LD50 orally in Rabbit: 1738 mg/kg LD50 dermal Rat > 2000 mg/kg |
| Hazard Information | Back Directory | [Chemical Properties]
colorless to yellow liquid | [Synthesis]
1,4-Butanediol vinyl ether is prepared by reacting acetylene and 1,4-butanediol in the presence of potassium hydroxide and potassium alkoxide catalyst: 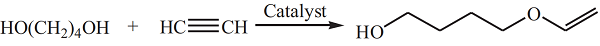 Since 1,4-butanediol contains two hydroxyl groups and the product contains active vinyl ether functional groups, some side reactions will occur. For example, further reaction of HBVE with acetylene to gen�erate divinyl ether, and self-condensation reaction of HBVE to form a cyclic acetal. A superbase catalytic system CsF–NaOH was developed for the preparation of HBVE at a temperature of 138–14 °C and under initial acetylene pressure of 1.0–1.2 MPa. The conversion of 1,4-butanediol was 100% and the total yield of the vinyl ethers was 80% at catalyst dosage (7 mol% CsF and 7 mol% NaOH based on 1,4-butanediol) and after reaction for 3 h. Some researchers also developed heterogeneous catalysts for the reaction of acetylene and 1,4-butanediol to obtain HBVE. Potassium hydroxide was supported on carriers such as aluminum oxide, molecular sieves, silica gel and zirconia.
Since 1,4-butanediol contains two hydroxyl groups and the product contains active vinyl ether functional groups, some side reactions will occur. For example, further reaction of HBVE with acetylene to gen�erate divinyl ether, and self-condensation reaction of HBVE to form a cyclic acetal. A superbase catalytic system CsF–NaOH was developed for the preparation of HBVE at a temperature of 138–14 °C and under initial acetylene pressure of 1.0–1.2 MPa. The conversion of 1,4-butanediol was 100% and the total yield of the vinyl ethers was 80% at catalyst dosage (7 mol% CsF and 7 mol% NaOH based on 1,4-butanediol) and after reaction for 3 h. Some researchers also developed heterogeneous catalysts for the reaction of acetylene and 1,4-butanediol to obtain HBVE. Potassium hydroxide was supported on carriers such as aluminum oxide, molecular sieves, silica gel and zirconia. | [Reactivity Profile]
The structure of the 1,4-Butanediol vinyl ether (HBVE) is a vinyl double bond directly connected with an ether bond, and due to the influence of an adjacent oxygen atom, the double bond is an electron-rich double bond and shows higher reactivity. HBVE also has a hydroxyl, and can react with a series of resins. It has wide application in fluororesin and coatings.
| [Flammability and Explosibility]
Flammable |
|
|