| Identification | Back Directory | [Name]
(1S)-1,5-Anhydro-1-C-[3-[(1-benzothiophen-2-yl)methyl]-4-fluorophenyl]-D-glucitol | [CAS]
761423-87-4 | [Synonyms]
CS-1580
Ipragliflozin
Ipragliflozin-002
Ipragliflozin, >=98%
Ipragliflozin impuity
IpragliflozinL-Proline
Ipragliflozin Impurity
Ipragliflozin (Suglat)
Ipragliflozin (ASP1941)
IpragliflozinL-Proline int
D-Glucitol, 1,5-anhydro-1-C-[3-(benzo[b]thien-2-ylmethyl)-4-fluorophenyl]-, (1S)-
(1S)-1,5-Anhydro-1-C-[3-[(1-benzothiophen-2-yl)methyl]-4-fluorophenyl]-D-glucitol
ASP-1941/D-Glucitol, 1,5-anhydro-1-C-[3-(benzo[b]thien-2-ylMethyl)-4-fluorophenyl]-, (1S)-
Ipragliflozin, (1S)-1,5-Anhydro-1-C-[3-[(1-benzothiophen-2-yl)Methyl]-4-fluorophenyl]-D-glucitol
(2S,3R,4R,5S,6R)-2-[3-(1-benzothiophen-2-ylmethyl)-4-fluorophenyl]-6-(hydroxymethyl)oxane-3,4,5-triol | [Molecular Formula]
C21H21FO5S | [MDL Number]
MFCD19443744 | [MOL File]
761423-87-4.mol | [Molecular Weight]
404.45 |
| Chemical Properties | Back Directory | [Melting point ]
155-157°C | [Boiling point ]
628.8±55.0 °C(Predicted) | [density ]
1.452 | [storage temp. ]
Refrigerator | [solubility ]
DMSO (Slightly), Methanol (Slightly) | [form ]
Solid | [pka]
13.27±0.70(Predicted) | [color ]
White to Off-White | [InChI]
InChI=1/C21H21FO5S/c22-15-6-5-12(21-20(26)19(25)18(24)16(10-23)27-21)7-13(15)9-14-8-11-3-1-2-4-17(11)28-14/h1-8,16,18-21,23-26H,9-10H2/t16-,18-,19+,20-,21+/s3 | [InChIKey]
AHFWIQIYAXSLBA-OPUVIHLBNA-N | [SMILES]
O1[C@H](CO)[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1C1C=CC(F)=C(CC2=CC3C=CC=CC=3S2)C=1 |&1:1,4,6,8,10,r| |
| Hazard Information | Back Directory | [Description]
Ipragliflozin L-proline was approved in Japan in January 2014
for the treatment of type 2 diabetes. The drug was discovered by
Astellas Pharma and co-developed and marketed with Kotobuki
Pharmaceutical and Merck Sharp Dohme as Suglat?. Similar to
empagliflozin (XIII), ipragliflozin L-proline is a sodium-glucose
1956 A. C. Flick et al. / Bioorg. Med. Chem. 24 (2016) 1937–1980
co-transporter-2 inhibitor which prevents glucose reabsorption by
excreting excess glucose in the urine. Ipragliflozin exhibits
remarkable selectivity over SLGT-1 (>250x). | [History]
Ipragliflozin L-proline was approved in Japan in January 2014 for the treatment of type 2 diabetes. The drug was discovered by Astellas Pharma and co-developed and marketed with Kotobuki Pharmaceutical and Merck Sharp Dohme as Suglat?. Similar to empagliflozin (XIII), ipragliflozin L-proline is a sodium-glucose 1956 A. C. Flick et al. / Bioorg. Med. Chem. 24 (2016) 1937–1980 co-transporter-2 inhibitor which prevents glucose reabsorption by excreting excess glucose in the urine. Ipragliflozin exhibits remarkable selectivity over SLGT-1 (>250x). | [Uses]
Ipragliflozin is a potent and selective inhibitor of sodium-glucose cotransporter-2 (SGLT2) and can serve as a potential agent for the treatment of type 1 and type 2 diabetes. | [Definition]
ChEBI:Ipragliflozin is a glycoside. | [Trade name]
Suglat | [Synthesis]
Commercial 5-bromo-2-fluorobenzaldehyde (123) was
subjected to nucleophilic attack upon subjection to lithiated
benzo[b]thiophene (124) to afford the dibenzylic alcohol 125 in
85% yield. This alcohol was then halogenated by means of thionyl
chloride in acetonitrile to give 126, which was followed by treatment
with sodium borohydride to give rise to 2-(5-bromo-2-fluorophenyl)-
1-benzothiophene (127), which was isolated by
crystallization from 2-propanol and methanol in 81% yield across
the two steps. Bromide 127 then underwent lithium¨Chalogen
exchange prior to exposure to 2,3,4,6-tetrakis-O-(trimethylsilyl)-
D-glucono-1,5-lactone (128) in toluene. Without workup, the
resulting mixture was treated with a solution of methanol and
HCl at 0 �C to give a globally desilylated a-glucopyranoside intermediate.
Subjection to acetic anhydride and 4-dimethylaminopyridine
furnished tetra-O-acetyl ipragliflozin (129) in 75% yield for
the 3 steps. Polyacetate 129 was then saponified using aqueous
sodium hydroxide and the product was crystallized from methanol
and water and subsequently treated with D-proline in ethanol to
furnish the desired product ipragliflozin D-proline (XVI) in 68%
yield.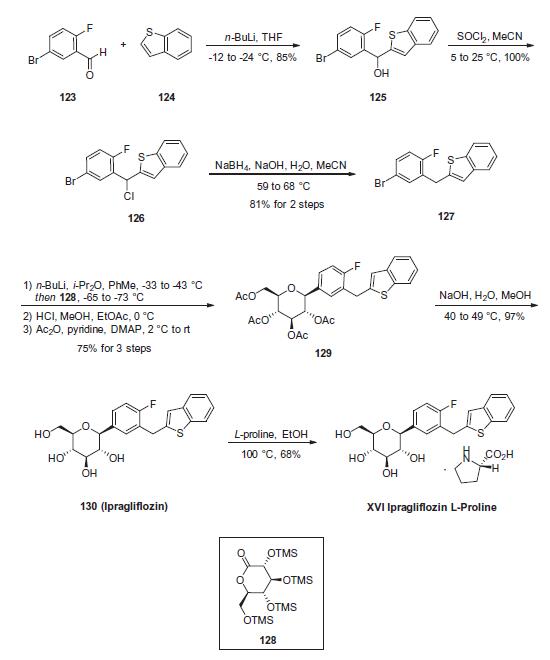
| [storage]
Store at -20°C | [Mode of action]
Ipragliflozin is a selective SGLT2 (sodium-glucose co-transporter 2) inhibitor discovered through research collaboration with Kotobuki Pharmaceutical Co., Ltd. SGLTs are membrane proteins that exist on the cell surface and transfer glucose into cells. SGLT2 is a subtype of the sodium-glucose co-transporters and plays a key role in the reuptake of glucose in the proximal tubule of the kidneys. Ipragliflozin reduces blood glucose levels by inhibiting the reuptake of glucose. | [References]
[1] Patent: EP2105442, 2009, A1. Location in patent: Page/Page column 13
[2] Patent: CN108276396, 2018, A. Location in patent: Paragraph 0041; 0052; 0053; 0068; 0083
[3] Bioorganic and Medicinal Chemistry, 2012, vol. 20, # 10, p. 3263 - 3279
[4] Patent: EP2009010, 2008, A1. Location in patent: Page/Page column 6-7 |
|
|