| Identification | More | [Name]
4-Isopropyltoluene | [CAS]
99-87-6 | [Synonyms]
1-ISOPROPYL-4-METHYLBENZENE
1-METHYL-4-ISOPROPYLBENZENE
4-CYMENE
4-ISOPROPYLTOLUENE
4-METHYLISOPROPYLBENZENE
CYMENE, 4-
CYMENE PARA
DOLCYMENE
FEMA 2356
METHYL(1-METHYLETHYL)BENZENE
Methyl isopropyl benzene
PARA CYMENE
PARA-CYMENE 97
P-CYMENE
P-CYMOL
P-ISOPROPYLTOLUENE
P-METHYLPROPYLBENZENE
1-(1-methylethyl)-4-methylbenzene
1-isopropyl-4-methyl-benzen
1-methyl-4-(1-methylethyl)-benzen | [EINECS(EC#)]
202-796-7 | [Molecular Formula]
C10H14 | [MDL Number]
MFCD00008893 | [Molecular Weight]
134.22 | [MOL File]
99-87-6.mol |
| Chemical Properties | Back Directory | [Appearance]
colourless liquid with a characteristic odour | [Melting point ]
-68 °C
| [Boiling point ]
176-178 °C(lit.)
| [density ]
0.86 g/mL at 25 °C(lit.)
| [vapor density ]
4.62 (vs air)
| [vapor pressure ]
1.5 mm Hg ( 20 °C)
| [FEMA ]
2356 | [refractive index ]
n20/D 1.490(lit.)
| [Fp ]
117 °F
| [storage temp. ]
Flammables area | [solubility ]
Chloroform (Sparingly), Ethyl Acetate (Slightly) | [form ]
Liquid | [color ]
Clear | [Odor]
Mild, pleasant; aromatic, solvent-type. | [Stability:]
Stable. Flammable. Incompatible with strong oxidizing agents, strong acids, strong bases. | [biological source]
synthetic | [explosive limit]
5.6% | [Odor Threshold]
0.057ppm | [Odor Type]
terpenic | [Water Solubility ]
PRACTICALLY INSOLUBLE | [Thermal Conductivity]
0.122 W/(m·K) at 25 ℃ | [JECFA Number]
1325 | [Merck ]
14,2763 | [BRN ]
1903377 | [Henry's Law Constant]
9.6×10-4 mol/(m3Pa) at 25℃, Plyasunov and Shock (2000) | [Dielectric constant]
2.3(17℃) | [Major Application]
cleaning products
cosmetics
environmental
flavors and fragrances
food and beverages
personal care | [Cosmetics Ingredients Functions]
FRAGRANCE | [InChI]
1S/C10H14/c1-8(2)10-6-4-9(3)5-7-10/h4-8H,1-3H3 | [InChIKey]
HFPZCAJZSCWRBC-UHFFFAOYSA-N | [SMILES]
CC(C)c1ccc(C)cc1 | [LogP]
4.1-4.8 at 20℃ | [Surface tension]
28.1mN/m at 20°C | [CAS DataBase Reference]
99-87-6(CAS DataBase Reference) | [NIST Chemistry Reference]
Benzene, 1-methyl-4-(1-methylethyl)-(99-87-6) | [EPA Substance Registry System]
99-87-6(EPA Substance) |
| Safety Data | Back Directory | [Symbol(GHS) ]
  
GHS02,GHS08,GHS09 | [Signal word ]
Danger | [Hazard statements ]
H226-H304-H361f-H411 | [Precautionary statements ]
P201-P202-P210-P273-P301+P310-P331 | [Hazard Codes ]
Xi | [Risk Statements ]
R10:Flammable.
R36/37/38:Irritating to eyes, respiratory system and skin . | [Safety Statements ]
S26:In case of contact with eyes, rinse immediately with plenty of water and seek medical advice .
S36:Wear suitable protective clothing . | [RIDADR ]
UN 2046 3/PG 3
| [WGK Germany ]
2
| [RTECS ]
GZ5950000
| [Autoignition Temperature]
817 °F | [TSCA ]
Yes | [REACH Registrations]
Active | [HazardClass ]
3 | [PackingGroup ]
III | [HS Code ]
29029090 | [Storage Class]
3 - Flammable liquids | [Hazard Classifications]
Acute Tox. 3 Inhalation
Aquatic Chronic 2
Asp. Tox. 1
Flam. Liq. 3
Repr. 2 | [Safety Profile]
Mildly toxic by
ingestion. Humans sustain central nervous
system effects at low doses. A skin irritant.
Flammable liquid. Explosion Hazard:
Slight in the form of vapor. To fight fire,
use foam, CO2, dry chemical. When heated
to decomposition it emits acrid smoke and
fumes. | [Hazardous Substances Data]
99-87-6(Hazardous Substances Data) | [Toxicity]
LD50 orally in rats: 4750 mg/kg (Jenner) |
| Raw materials And Preparation Products | Back Directory | [Raw materials]
(+)-Dipentene-->1,8-Cineole-->Cyclohexane, methyl-, radical ion(1+)-->Bicyclo[3.1.0]hexan-3-ol, 4-methylene-1-(1-methylethyl)-, (1R,3S,5R)-rel--->Bicyclo[3.1.0]hex-2-ene,4-methylene-1-(1-methylethyl)--->CIS-1-ISOPROPYL-4-METHYLCYCLOHEXANE | [Preparation Products]
Acetone-->m-Cresol-->Cresol-->p-Toluic acid-->Isoproturon-->4-Bromomethylbenzoic acid-->Cuminaldehyde-->GAMMA-TERPINENE-->2-NITRO-4-CYMENE-->4-Isopropylbenzoic acid-->2-Chloro-1-methyl-4-(1-methylethyl)benzene-->p-menthatriene,p-mentha-1,3,8-triene-->6,6-Dimethyl-4-methylenebicyclo[3.1.1]hept-2-ene |
| Hazard Information | Back Directory | [General Description]
Colorless liquid with a mild pleasant odor. Floats on water. | [Reactivity Profile]
Vigorous reactions, sometimes amounting to explosions, can result from the contact between aromatic hydrocarbons, such as CYMENE, and strong oxidizing agents. They can react exothermically with bases and with diazo compounds. Substitution at the benzene nucleus occurs by halogenation (acid catalyst), nitration, sulfonation, and the Friedel-Crafts reaction. | [Air & Water Reactions]
Insoluble in water. | [Health Hazard]
Inhalation causes impairment of coordination, headache. Contact with liquid causes mild irritation of eyes and skin. Ingestion causes irritation of mouth and stomach. | [Fire Hazard]
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water. | [Occurrence]
Widespread in nature; reported in the oils of cypress, Artemisia cina Bg., Ceylon cinnamon, boldo leaf, cascarilla,
Cuminum cyminum L. and lemon; in the oils for seeds of coriander and Cicuta virosa; in the ether-extracted oils of Monarda
fisulosa and Monarda puncata; in the star anise and nutmeg essential oils. Note that p-cymene reported in essential oil may be
formed from the conversion of a cyclic terpene; its presence is indicative of aging in lemon essential oil. Reported found in citrus
juices and peel oils, guava, papaya, black currant, pineapple, blueberry, blackberry, carrot, tomato, celery seed, cumin seed, mace,
ginger, coriander seed, anise, nutmeg, mace, parsley, cardamom, cinnamon root and bark, pepper, peppermint oil, spearmint oil,
cassia leaf and Thymus vulgaris. | [Definition]
ChEBI: A monoterpene that is toluene substituted by an isopropyl group at position 4. | [Preparation]
Obtained chiefly from the wash water of sulfite paper. | [Aroma threshold values]
Aroma characteristics at 1% EtoH: harsh chemical, woody and terpy-like with an oxidized citrus lemon
note. It has spicy nuances reminiscent of cumin, oregano and cilantro | [Taste threshold values]
Taste characteristics at 1 to 10 ppm: terpy and rancid with slightly woody, oxidized citrus notes. It has spice
nuances of green pepper and oregano. | [Synthesis Reference(s)]
Journal of the American Chemical Society, 95, p. 982, 1973 DOI: 10.1021/ja00784a090 | [Flammability and Explosibility]
Flammable | [Chemical Reactivity]
Reactivity with Water: No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent. | [Metabolism]
In this compound both an isopropyl and a methyl group occur together and the available evidence shows that only the methyl group is oxidized, cumic acid (p�isopropylbenzoic acid) and its conjugate (cuminuric acid) being the main metabolites in dogs and in sheep (Williams, 1959). Following inhalation, only a small part is excreted un�changed, the remainder being oxidized to water-soluble metabolites. As early as 1873, Ziegler suggested that the readily oxidized propyl side-chain formed a -COO H group (Browning, 1965): The ultimate product in the case of dogs and sheep is cumic acid, which is probably excreted as a conjugate with glycine (Gerarde, 1960). | [Purification Methods]
Wash p-cymene with cold, conc H2SO4 until there is no further colour change, then repeatedly with H2O, 10% aqueous Na2CO3 and H2O again. Dry it over Na2SO4, CaCl2 or MgSO4, and distil it. Further purification steps include steam distillation from 3% NaOH, percolation through silica gel or activated alumina, and a preliminary reflux for several days over powdered sulfur. Store it over CaH2. [Beilstein 5 IV 1060.] | [Toxics Screening Level]
he screening level can be calculated from this LD50 value using
R232(1)(h) as follows. The initial threshold screening level (ITSL) for p-Cymene is 10 μg/m3 with annual averaging. |
| Questions And Answer | Back Directory | [Description]
p-Cymene is a naturally occurring compound, although it has two further geometric isomers, m-cymene (with meta-substituted alkyl groups) and o-cymene (with ortho-substituted alkyl groups) which do not occur naturally. p-Cymene is related to the monoterpenes. Monoterpenes themselves belong to the “terpenes”, a larger class of organic compounds, which are the most representative components of essential oils. chemical structure of p-cymene, indicating the benzene ring with methyl and isopropyl substitutions.
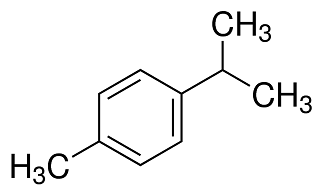
chemical structure of p-cymene
| [Chemical Properties]
p-Cymene has a citrusy aroma reminiscent of lemon. It is colorless liquid with a strong, characteristic odor reminiscent of carrot. It tends to darken with aging. It occurs naturally in orange peel oil, mandarin peel oil, satsuma mandarin peel oil, coldpressed lime peel oil, distilled lime peel oil, cinnamon leaf, etc. 4-isopropyltoluene has antimicrobial properties. It is used as a flavor ingredient permitted for direct addition to food for human consumption.
| [Uses]
p-Cymene is an alkyl aromatic hydrocarbon mainly used to study oxidation reactions under various conditions to synthesize highly valuable intermediates and building-blocks such as cymene hydroperoxide, 4-methyl acetophenone, 4-isopropyl benzyl alcohol, 4-isopropyl benzaldehyde and 4-isopropyl benzoic acid etc.
Medicinally, p-cymene is used to prevent coughs and eliminate phlegm as well as being a flavouring agent, and being used in the production of fungicides and pesticides. It is considered to be “generally recognised as safe” (GRAS) by the U.S. Food and Drug Administration. Its potential in the prevention of protein glycation mediated diabetic complications has also been confirmed, and it has been suggested as an in vivo antioxidant compound due to its ability to reduce the formation of oxygen and nitrogen reactive species, acting as a potential neuroprotective agent in the brain. Therefore, it could be involved in the treatment of oxidative stress related diseases.
| [Production Methods]
p-cymene is conventionally produced by the Friedel-Crafts alkylation of toluene with isopropanol, or of benzene with methyl or isopropyl halides.
| [References]
[1] George A. Burdock, Encyclopedia of Food and Color Additives, Volume 1, 1996
[2] C.F. Bagamboula, M. Uyttendaele and J. Debevere, Inhibitory effect of thyme and basil essential oils, carvacrol, thymol, estragol, linalool and p-cymene towards Shigella sonnei and S. flexneri, Food Microbiology, 2004, vol. 21, 33-42
|
|
|