18598-63-5
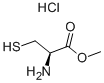 18598-63-5 结构式
18598-63-5 结构式
基本信息
半胱氨酸甲酯盐酸盐
L-盐酸半胱氨酸甲酯
半胱氨酸甲酯盐酸酯
L-半胱氨酸甲醇酯延酸盐
H-CYS-OME HCL
L-CYSTEINE METHYL ESTER HYDROCHLORIDE
L-CYSTEINE METHYL ESTER MONOHYDROCHLORIDE
L-CYSTEIN METHYL ESTER HYDROCHLORIDE
MECYSTEINE HYDROCHLORIDE
methyl cysteine hydrochloride
acdrile
actiol
l.j.48
methylester,hydrochloride,l-cystein
pectite
visclair
zeotin
L-Cysteine Methyl Ester HCl
L-CYSTEINE METHYL ESTER HYDROCHLORIDECRY STALLINE
L-CYSTEINE METHYL ESTER HYDROCHLORIDE, 9 8%
Mecysteine HCL
L-Cystein methyl ester HCl
H-CYS(ME)-OH . HCL
物理化学性质
安全数据
制备方法
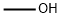
67-56-1
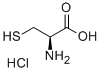
52-89-1

18598-63-5
在冰浴条件下,于氮气保护下,将3 mL的氯化亚砜(SOCl2)缓慢滴加至35 mL甲醇中。随后,将反应混合物于室温下搅拌1小时。之后,分批加入1 g L-半胱氨酸盐酸盐一水合物(Cys·HCl·H2O)至上述溶液中。将混合溶液在室温下继续反应3小时,然后加热回流1小时。反应完成后,通过减压蒸馏除去挥发性组分及溶剂,所得残余物用甲醇-二氯甲烷(CH3OH-CH2Cl2)混合溶剂进行重结晶,得到0.84 g白色固体产物L-半胱氨酸甲酯盐酸盐,产率为85.9%。产物经红外光谱(IR, KBr压片)分析显示特征吸收峰:3040.4 cm-1(强,NH2伸缩振动),1709.7 cm-1(强,C=O伸缩振动),2580 cm-1(弱,SH伸缩振动)。核磁共振氢谱(1H NMR, D2O为溶剂,TMS为内标)数据如下:δ 4.453 (t, 1H, CH, J = 5.2 Hz), 3.871 (s, 3H, CH3), 3.164 (t, 2H, CH2, J = 6.4 Hz)。元素分析结果:计算值(C4H10NO2SCCl): C, 27.99%; H, 5.87%; N, 8.16%。实测值:C, 28.11%; H, 6.01%; N, 8.08%。
参考文献:
[1] European Journal of Medicinal Chemistry, 2014, vol. 74, p. 199 - 215
[2] Journal of Organic Chemistry, 2013, vol. 78, # 9, p. 4270 - 4277
[3] Tetrahedron Asymmetry, 2000, vol. 11, # 21, p. 4255 - 4261
[4] Journal of the American Chemical Society, 2008, vol. 130, # 15, p. 5052 - 5053
[5] European Journal of Medicinal Chemistry, 2012, vol. 55, p. 176 - 187,12
