25122-57-0
中文名称
丁酸氯倍他松
英文名称
Clobetasone butyrate
CAS
25122-57-0
EINECS 编号
246-635-9
分子式
C22H26ClFO4
MDL 编号
MFCD00133196
分子量
408.891
MOL 文件
25122-57-0.mol
更新日期
2026/05/19 12:26:32
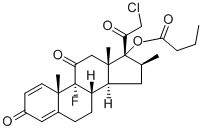 25122-57-0 结构式
25122-57-0 结构式
基本信息
中文别名
丁酸倍氯他松丁酸氯倍他松
氯倍他松丁酸酯
英文别名
(16-beta)-)11,20-trione,21-chloro-9-fluoro-17-hydroxy-16-beta-methyl-pregna-4-diene-3
21-chloro-9-fluoro-17-hydroxy-16-beta-methylpregna-1,4-diene,3,11,20-trione
eumovate
molivate
pregna-1,4-diene-3,11,20-trione,21-chloro-9-fluoro-16-methyl-17-(1-oxobutoxy
sn203
21-chloro-9-fluoro-17-hydroxy-16beta-methylpregna-1,4-diene-3,11,20-trione 17-butyrate
ClobetasolPropionate-Bp/Ep/Usp
Butyric acid, ester with 21-chloro-9-fluoro-17-hydroxy-16β-methylpregna-1,4-diene-3,11,20-trione (8CI)
CCl 5537
Emovate
GR 2/1214
Kindavate
Pregna-1,4-diene-3,11,20-trione, 21-chloro-9-fluoro-16-methyl-17-(1-oxobutoxy)-, (16β)-
Pregna-1,4-diene-3,11,20-trione, 21-chloro-9-fluoro-17-hydroxy-16β-methyl-, butyrate (8CI)
21-Chloro-9-fluoro-17-hydroxy-16b-methylpregna-1,4-diene-3,11,20-trione 17-butyrate
Clobetasone butyrate
Clobetasone-17-butyrate
CLOBETASOLE BUTYRATE
所属类别
分析化学:药典标准品和杂质标准品物理化学性质
熔点90-100°
沸点573.3±50.0 °C(Predicted)
密度1.1736 (estimate)
储存条件Refrigerator, Under Inert Atmosphere
溶解度Practically insoluble in water, freely soluble in acetone and in methylene chloride, slightly soluble in ethanol (96 per cent).
形态neat
颜色白色至灰白色
稳定性稳定的。易燃。与强氧化剂不相容。
InChIKeyFBRAWBYQGRLCEK-JGSYYNBSNA-N
SMILES[C@]1(C(=O)CCl)(OC(=O)CCC)[C@@H](C)C[C@@]2([H])[C@]3([H])CCC4=CC(=O)C=C[C@]4(C)[C@@]3(F)C(=O)C[C@]12C |&1:0,11,14,16,26,28,33,r|
常见问题列表
用途
丁酸氯倍他松是一种外用糖皮质激素药,具有较强的毛细血管收缩作用,临床上主要用于治疗皮肤病。
生物活性
Clobetasone butyrate是一种新型的皮质类固醇,经常用于治疗湿疹、牛皮癣、皮肤炎和某些眼科疾病。靶点
IC50: glucocorticoid
体内研究
Clobetasone butyrate (application on the skin; 0.05%; 7 weeks) induces epidermal thinning in skin, Clobetasone butyrate 7 weeks causes much less epidermal thinning in the pig than flurandrenolone (0.05%) or fluocinolone acetonide (0.025%), both of which causes severe atrophy.
| Animal Model: | Large white domestic pig |
| Dosage: | 0.5% application on the skin |
| Administration: | 7 weeks |
| Result: | Caused moderate to slight atrophy. |
