N-OCTANE
- CAS No.
- 111-65-9
- Chemical Name:
- N-OCTANE
- Synonyms
- OCTANE;Octan;1-Octane;Oatane;octanes;Oktan;Ottani;n-C8H18;n-Octan;Oktanen
- CBNumber:
- CB2124951
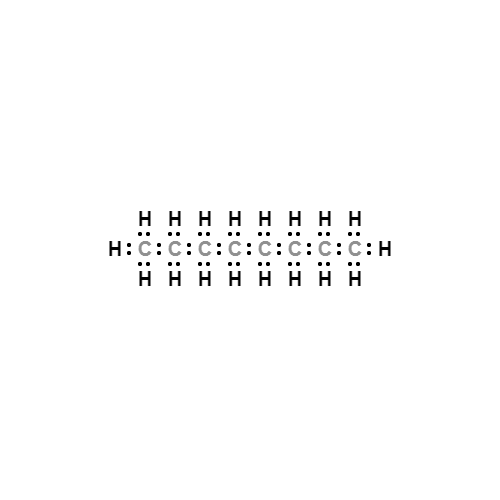
- Molecular Formula:
- C8H18
Lewis structure
- Molecular Weight:
- 114.23
- MDL Number:
- MFCD00009556
- MOL File:
- 111-65-9.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| n-Octane for synthesis | 8.06910 | 100mL | $56.5 |
| n-Octane for synthesis | 8.06910 | 250ML | $84.1 |
| n-Octane for synthesis | 8.06910 | 1L | $264 |
| Octane anhydrous, ≥99% | 296988 | 1l | $422 |
| Octane anhydrous, ≥99% | 296988 | 4x2l | $1980 |
| More product size | |||
| Melting point | −57 °C(lit.) |
|---|---|
| Boiling point | 125-127 °C(lit.) |
| Density | 0.703 g/mL at 25 °C(lit.) |
| vapor density | 3.9 (vs air) |
| vapor pressure | 11 mm Hg ( 20 °C) |
| refractive index |
n |
| Flash point | 60 °F |
| storage temp. | Store below +30°C. |
| solubility | ethanol: soluble(lit.) |
| form | Liquid |
| pka | >14 (Schwarzenbach et al., 1993) |
| color | Clear colorless |
| Odor | Like gasoline. |
| explosive limit | 0.8-6.5%(V) |
| Odor Threshold | 1.7ppm |
| Water Solubility | 0.0007 g/L (20 ºC) |
| Thermal Conductivity | 0.1244 W/(m·K) at 25 ℃ |
| Merck | 14,6749 |
| BRN | 1696875 |
| Henry's Law Constant | 4.45 at 25 °C (Jnsson et al., 1982) |
| Exposure limits | TLV-TWA 300 ppm (~1450 mg/m3) (ACGIH and NIOSH), 500 ppm (~2420 mg/m3) (OSHA); STEL 375 ppm (~1800 mg/m3). |
| Dielectric constant | 1.1(-4℃) |
| Stability | Stable. Highly flammable. Readily forms explosive mixtures with air. Incompatible with oxidizing agents. |
| Cosmetics Ingredients Functions | SOLVENT |
| InChI | 1S/C8H18/c1-3-5-7-8-6-4-2/h3-8H2,1-2H3 |
| InChIKey | TVMXDCGIABBOFY-UHFFFAOYSA-N |
| SMILES | CCCCCCCC |
| LogP | 5.15 |
| Surface tension | 21.1mN/m at 298.15K |
| CAS DataBase Reference | 111-65-9(CAS DataBase Reference) |
| EWG's Food Scores | 1-2 |
| FDA UNII | X1RV0B2FJV |
| NIST Chemistry Reference | Octane(111-65-9) |
| EPA Substance Registry System | Octane (111-65-9) |
| UNSPSC Code | 41116107 |
| NACRES | NA.24 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |     GHS02,GHS07,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H304-H315-H336-H410 | |||||||||
| Precautionary statements | P210-P233-P273-P301+P310-P303+P361+P353-P331 | |||||||||
| target organs | Central nervous system | |||||||||
| Hazard Codes | F,Xn,N | |||||||||
| Risk Statements | 11-38-50/53-65-67 | |||||||||
| Safety Statements | 9-16-29-33-60-61-62 | |||||||||
| OEB | A | |||||||||
| OEL | TWA: 75 ppm (350 mg/m3), Ceiling: 385 ppm (1800 mg/m3) [15-minute] | |||||||||
| RIDADR | UN 1262 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | RG8400000 | |||||||||
| Autoignition Temperature | 428 °F | |||||||||
| TSCA | TSCA listed | |||||||||
| HazardClass | 3 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29011000 | |||||||||
| Storage Class | 3 - Flammable liquids | |||||||||
| Hazard Classifications | Aquatic Acute 1 Aquatic Chronic 1 Asp. Tox. 1 Flam. Liq. 2 Skin Irrit. 2 STOT SE 3 |
|||||||||
| Hazardous Substances Data | 111-65-9(Hazardous Substances Data) | |||||||||
| Toxicity | LDLo intravenous in mouse: 428mg/kg | |||||||||
| IDLA | 1,000 ppm [10% LEL] | |||||||||
| Limited Quantities | 1.0 L (0.3 gallon) (liquid) | |||||||||
| Excepted Quantities | Max Inner Pack (30g or 30ml) and Max Outer Pack (500g or 500ml) | |||||||||
| NFPA 704 |
|
N-OCTANE price More Price(42)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 8.06910 | n-Octane for synthesis | 111-65-9 | 100mL | $56.5 | 2026-04-30 | Buy |
| Sigma-Aldrich | 8.06910 | n-Octane for synthesis | 111-65-9 | 250ML | $84.1 | 2026-04-30 | Buy |
| Sigma-Aldrich | 8.06910 | n-Octane for synthesis | 111-65-9 | 1L | $264 | 2026-04-30 | Buy |
| Sigma-Aldrich | 296988 | Octane anhydrous, ≥99% | 111-65-9 | 1l | $422 | 2026-04-30 | Buy |
| Sigma-Aldrich | 296988 | Octane anhydrous, ≥99% | 111-65-9 | 4x2l | $1980 | 2026-04-30 | Buy |
N-OCTANE Chemical Properties,Uses,Production
Description
Octane is a colorless liquid with a gasolinelike odor. The odor threshold is 4 ppm and 48 ppm (NewJersey Fact Sheet). Molecular weight = 114.26; Specificgravity (H2O:1)= 0.70; Boiling point = 125.6℃; Freezing/Melting point = 256.7℃; Vapor pressure =10 mmHg at20℃; Flash point =13℃(cc); Autoignition temperature =206℃. Explosive limits: LEL 5 1.0%; UEL = 6.5%.Hazard Identification (based on NFPA-704 M RatingSystem): Health 0, Flammability 3, Reactivity 0. Practicallyinsoluble in water; solubility = 7 × 10-5.
Chemical Properties
colourless liquid
Chemical Properties
Octane is a colorless liquid with a gasoline-like odor. The odor threshold is 4 ppm and 48 ppm (New Jersey Fact Sheet).
Physical properties
Clear, colorless, flammable liquid with a gasoline-like odor. An odor threshold concentration of 1.7 ppmv was reported by Nagata and Takeuchi (1990).
Uses
n-Octane occurs in petroleum crackingproducts, gasoline, petroleum ether, andpetroleum naphtha. It is used as a solventand in organic synthesis.
Uses
As a constituent in motor and aviation fuels; as an industrial solvent; in organic synthesis
Uses
n-Octane is used as a solvent and raw material for organic synthesis reactions and is a very important chemical in the petroleum industry. It is also widely used in the rubber and paper processing industries. Isooctane, along with other nalkanes and isoparaffins, is used in the blending of fuels to achieve desired antiknock properties.
Production Methods
Octane is produced from the fractional distillation and refining of petroleum.
Definition
A liquid alkane obtained from the light fraction of crude oil. Octane and its isomers are the principal constituents of gasoline, which is obtained as the refined light fraction from crude oil.
Definition
ChEBI: A straight chain alkane composed of 8 carbon atoms.
Synthesis Reference(s)
The Journal of Organic Chemistry, 55, p. 6194, 1990 DOI: 10.1021/jo00312a029
Tetrahedron, 48, p. 8881, 1992 DOI: 10.1016/S0040-4020(01)81987-6
Tetrahedron Letters, 31, p. 5093, 1990 DOI: 10.1016/S0040-4039(00)97814-6
General Description
Colorless liquid with an odor of gasoline. Less dense than water and insoluble in water. Hence floats on water. Produces irritating vapor.
Air & Water Reactions
Highly flammable. Insoluble in water.
Reactivity Profile
May be incompatible with strong oxidizing agents like nitric acid. Charring may occur followed by ignition of unreacted material and other nearby combustibles. In other settings, mostly unreactive. Not affected by aqueous solutions of acids, alkalis, most oxidizing agents, and most reducing agents. When heated sufficiently or when ignited in the presence of air, oxygen or strong oxidizing agents, burns exothermically to produce mostly carbon dioxide and water.
Health Hazard
Inhalation of concentrated vapor may cause irritation of respiratory tract, depression, and pulmonary edema. Liquid can cause irritation of eyes and (on prolonged contact) irritation and cracking of skin. Ingestion causes irritation of mouth and stomach. Aspiration causes severe lung irritation, rapidly developing pulmonary edema, and central nervous system excitement, followed by depression.
Health Hazard
The toxic properties of n-octane are similarto those of other paraffinic hydrocarbons. Itis an irritant to mucous membranes, and athigh concentrations it shows narcotic actions.The narcotic concentrations in mice werereported to be 8000–10,000 ppm (Patty andYant 1929) and the fatal concentration was13,500 ppm (Flury and Zernick 1931). Deathoccurred from respiratory arrest. The acutetoxicity of n-octane is somewhat greater thanthat of n-heptane.
Fire Hazard
Behavior in Fire: Vapor is heavier than air and may travel a considerable distance to a source of ignition and flash back.
Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
Safety Profile
Poison by intravenous route. May act as a simple asphyxiant. See also ARGON for a description of simple asphyxiants. A narcotic in high concentration. Human dermal exposure to undiluted octane for five hours resulted in blister formation but no anesthesia; exposure for one hour caused diffuse burning sensation. A very dangerous fire hazard and severe explosion hazard when exposed to heat, flame, or oxidizers. When heated to decomposition it emits acrid smoke and irritating fumes. See also ALKANES.
Potential Exposure
Octane is used as a solvent; as a fuel; as an intermediate in organic synthesis; and in azeotropicdistillations.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Do NOT induce vomiting. Give victim nothing to drink.Note to physician: Inhalation: bronchodilators, decongestants, and oxygen may be used if necessary.Corticosteroids are useful for treating pneumonitis.
Carcinogenicity
The promoting activity of octane in skin carcinogenesis, including its physical effect on micellar models of biological membranes, was tested. Octane proved to have significant promoting activity when tested as a 75% solution in cyclohexane.
Source
Schauer et al. (1999) reported octane in a diesel-powered medium-duty truck exhaust at
an emission rate of 260 μg/km.
Identified as one of 140 volatile constituents in used soybean oils collected from a processing plant that fried various beef, chicken, and veal products (Takeoka et al., 1996).
Schauer et al. (2001) measured organic compound emission rates for volatile organic
compounds, gas-phase semi-volatile organic compounds, and particle-phase organic compounds
from the residential (fireplace) combustion of pine, oak, and eucalyptus. The gas-phase emission
rate of octane was 1.7 mg/kg of pine burned. Emission rates of octane were not measured during
the combustion of oak and eucalyptus.
California Phase II reformulated gasoline contained octane at a concentration of 6.38 g/kg. Gasphase
tailpipe emission rates from gasoline-powered automobiles with and without catalytic
converters were 1.07 and 131 mg/km, respectively (Schauer et al., 2002).
Environmental Fate
Biological. n-Octane may biodegrade in two ways. This first is the formation of octyl
hydroperoxide, which decomposes to 1-octanol followed by oxidation to octanoic acid. The other
pathway involves dehydrogenation to 1-octene, which may react with water giving 1-octanol
(Dugan, 1972). 1-Octanol was reported as the biodegradation product of octane by a Pseudomonas
sp. (Riser-Roberts, 1992). Microorganisms can oxidize alkanes under aerobic conditions (Singer
and Finnerty, 1984). The most common degradative pathway involves the oxidation of the
terminal methyl group forming the corresponding alcohol (1-octanol). The alcohol may undergo a
series of dehydrogenation steps forming an aldehyde (octanal) then a fatty acid (octanoic acid).
The fatty acid may then be metabolized by β-oxidation to form the mineralization products,
carbon dioxide and water (Singer and Finnerty, 1984).
Photolytic. The following rate constants were reported for the reaction of octane and OH
radicals in the atmosphere: 5.1 x 10-12 cm3/moleculesec at 300 K (Hendry and Kenley, 1979); 1.34
x 10-12 cm3/moleculesec (Greiner, 1970); 8.40 x 10-12 cm3/moleculesec (Atkinson et al., 1979),
8.42 x 10-12 cm3/moleculesec at 295 K (Darnall et al., 1978). Photooxidation reaction rate
constants of 8.71 x 10-12 and 1.81 x 10-18 cm3/moleculesec were reported for the reaction of octane
with OH and NO3, respectively (Sablji and Güsten, 1990).
Surface Water. Mackay and Wolkoff (1973) estimated an evaporation half-life of 3.8 sec from a
surface water body that is 25 °C and 1 m deep.
Chemical/Physical. Complete combustion in air produces carbon dioxide and water vapor.
Octane will not hydrolyze because it does not contain a hydrolyzable functional group.
storage
Color Code—Red: Flammability Hazard: Store ina flammable liquid storage area or approved cabinet awayfrom ignition sources and corrosive and reactive materials.Prior to working with this chemical you should be trainedon its proper handling and storage. Before entering confinedspace where this chemical may be present, check to makesure that an explosive concentration does not exist. Octanemust be stored to avoid contact with strong oxidizers (suchas chlorine and bromine) because violent reactions occur.Store in tightly closed containers in a cool, well-ventilatedarea away from heat. Sources of ignition, such as smokingand open flames, are prohibited where octane is used, handled, or stored. Metal containers used in the transfer of 5gallons or more of octane should be grounded and bonded.Drums must be equipped with self-closing valves, pressurevacuum bungs, and flame arresters. Use only nonsparkingtools and equipment, especially when opening and closingcontainers of octane.
Shipping
UN1262 Octanes, Hazard Class: 3; Labels: 3-Flammable liquid.
Purification Methods
Extract the octane repeatedly with conc H2SO4 or chlorosulfonic acid, then wash it with water, dry and distil it. Alternatively, purify it by azeotropic distillation with EtOH, followed by washing with water to remove the EtOH, drying and distilling it. For further details, see n-heptane. It is also purified by zone melting. [Beilstein 1 H 159, 1 I 60, 1 II 122, 1 III 457, 1 IV 412.]
Toxicity evaluation
The mechanism of toxicity is suspected to be similar to other
solvents that rapidly induce anesthesia-like effects, i.e.,
a ‘nonspecific narcosis’ due to disruption (solvation) of the
integrity of the cellular membranes of the central nervous
system (CNS).
Octane is generally considered to be relatively nontoxic
relative to the effect seen following exposure to other aliphatic
hydrocarbons. This is probably due to the fact that it is less
volatile than the shorter chain aliphatic hydrocarbons (e.g.,
pentane or heptane) and may not be as readily transferred
across either the pulmonary alveoli or the blood–brain barrier.
If it is aspirated into the lungs, however, n-octane will cause
adverse effects similar to effects seen following aspiration of
other petroleum distillates or compounds.
Incompatibilities
Reacts with strong oxidizers, causing fire and explosion hazard. Attacks some forms of plastics, rubber and coatings.
Waste Disposal
Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an after burner and scrubber. All federal, state, and local environmental regulations must be observed.
N-OCTANE Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
1of2
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| BEYOND INDUSTRIES (CHINA) LIMITED | +86-21-52699951; +8613917686115 | sales@beyondindustriesgroup.com | China | 699 | 58 |
| Capot Chemical Co.,Ltd. | +86-(0)57185586718; +8613336195806 | sales@capot.com | China | 29640 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21588 | 55 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49977 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29794 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34563 | 58 |
| ANHUI WITOP BIOTECH CO., LTD | +8615255079626 | eric@witopchemical.com | China | 23541 | 58 |
| AFINE CHEMICALS LIMITED | +86-0571-85134551 | sales@afinechem.com | China | 15082 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +8617792793228 | 1069@dideu.com | China | 8401 | 58 |
| Hebei Fengjia New Material Co., Ltd | +86-0311-87836622 +86-18712993135 | sales01@tairunfaz.com | China | 8051 | 58 |
View Lastest Price from N-OCTANE manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-04-22 | n-Octane
111-65-9
|
US $0.00 / kg | 200kg | 99% | 20 tons | Qingdao RENAS Polymer Material Co., Ltd. | |
 |
2023-07-27 | N-OCTANE
111-65-9
|
US $1.10 / g | 1g | 99.0% Min | 100 Tons | Shaanxi Didu New Materials Co. Ltd | |
 |
2020-01-13 | N-OCTANE
111-65-9
|
US $1.00 / KG | 1KG | 98%-99.9% | 200kg | Career Henan Chemical Co |