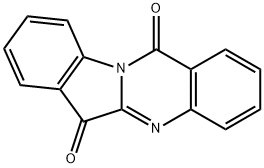
TRYPTANTHRIN
- CAS No.
- 13220-57-0
- Chemical Name:
- TRYPTANTHRIN
- Synonyms
- Couroupitine A;INDOLO[2,1-B]QUINAZOLINE-6,12-DIONE;NSC 349447;Trypanthrine;TRYPTANTHRIN;tryptanthrine;TRYPTANTHRIN-D4;Color amine ketone;6,12-Dihydroindolo[2,1-b]quinazoline-6,12-dione;CouroupitineAIndolo(2,1-b)quinazoline-6,12-dioneTryptanthrine
- CBNumber:
- CB4477493
- Molecular Formula:
- C15H8N2O2
- Molecular Weight:
- 248.24
- MDL Number:
- MFCD00012073
- MOL File:
- 13220-57-0.mol
- MSDS File:
- SDS
| Melting point | 258 °C(Solv: ethanol (64-17-5)) |
|---|---|
| Boiling point | 469.3±28.0 °C(Predicted) |
| Density | 1.45±0.1 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| solubility | DMSO: soluble2mg/mL (warmed) |
| pka | -3.06±0.20(Predicted) |
| form | powder |
| color | faint yellow to dark yellow |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
| InChIKey | VQQVWGVXDIPORV-UHFFFAOYSA-N |
| FDA UNII | 4Y6E3F2U66 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302 |
| Precautionary statements | P301+P312+P330 |
| WGK Germany | 3 |
| HS Code | 29339900 |
TRYPTANTHRIN price More Price(28)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | SML0310 | Tryptanthrin ≥98% (HPLC) | 13220-57-0 | 5mg | $78.1 | 2024-03-01 | Buy |
| Sigma-Aldrich | SML0310 | Tryptanthrin ≥98% (HPLC) | 13220-57-0 | 25mg | $252 | 2024-03-01 | Buy |
| Cayman Chemical | 17913 | Tryptanthrin ≥95% | 13220-57-0 | 1mg | $32 | 2024-03-01 | Buy |
| Cayman Chemical | 17913 | Tryptanthrin ≥95% | 13220-57-0 | 5mg | $63 | 2024-03-01 | Buy |
| Sigma-Aldrich | PHL83458 | Tryptanthrine phyproof Reference Substance | 13220-57-0 | 10mg | $430 | 2024-03-01 | Buy |
TRYPTANTHRIN Chemical Properties,Uses,Production
Description
The matured fruit of Couroupita guianensis yields two highly unsaturated alkaloids. Couroupitine A is obtained in the form of yellow needles when crystallized from MeOH-CHCI3. It gives an ultraivolet spectrum with absorption maxima at 225 and 251 nm and an inflexion at 315 nm in EtOH.
Uses
Tryptanthrin is natural alkaloid, and a potent inhibitor of cyclooxygenase COX-2 and 5-lipoxygenase acting as an anti-inflammatory agent.
Definition
ChEBI: Tryptanthrine is an organonitrogen heterocyclic compound, an organic heterotetracyclic compound and an alkaloid antibiotic.
General Description
This substance is a primary reference substance with assigned absolute purity (considering chromatographic purity, water, residual solvents, inorganic impurities). The exact value can be found on the certificate. Produced by PhytoLab GmbH & Co. KG
Biochem/physiol Actions
Tryptanthrin is a plant alkaloid with anti-inflammatory and anti cancer activities. Tryptanthrin blocks leukotriene production in neutrophils and in whole blood assays, and in an in vivo rat pleurisy model. The compound also inhibits P-glycoprotein, and sensitizes resistant cancer cell lines to killing by cytotoxic agents.
in vitro
in human hepatocyte-derived hepg2 cells, tryptanthrin inhibited the reactive oxygen species formation, mitochondrial dysfunction, and cell death triggered by tert-butyl hydroperoxide (tbhp). furthermore, tryptanthrin reversed the reduction of glutathione (gsh) induced by tbhp. specifically, nuclear translocation, transactivation of nuclear factor erythroid 2-related factor 2 (nrf2), and phosphorylation of extracellular signal-regulated kinase (erk) were evoked by the treatment of tryptanthrin. additionally, the expression of the heme oxygenase 1 and glutamate was upregulated by tryptanthrin [1].
in vivo
balb/c (h-2d) mice, intraperitoneally (i.p.) inoculated with leukemia wehi-3b jcs cells, were injected i.p with tryptanthrin at doses of 0.04 mg/kg, 0.08 mg/kg and 0.16 mg/kg body weight for 5 consecutive days. tryptanthrin inhibited the growth of wehi-3b jcs cells in balb/c mice, and at the dosages of 0.08 mg/kg and 0.16 mg/kg, significant inhibition was seen. moreover, tryptanthrin, in a dose-dependent fashion, triggered cell cycle arrest of the wehi-3b jcs cells at g0/g1 phase [2].
References
Sen, Mahato, Dutta, Tetrahedron Lett., 609 (1974)
TRYPTANTHRIN Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Accela ChemBio Inc. | +1-858-6993322 | info@accelachem.com | United States | 19536 | 58 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 | linda@hubeijusheng.com | CHINA | 22968 | 58 |
| Alchem Pharmtech,Inc. | 8485655694 | sales@alchempharmtech.com | United States | 63711 | 58 |
| Fuxin Pharmaceutical | +86-021-021-50872116 +8613122107989 | contact@fuxinpharm.com | China | 10297 | 58 |
| Zhengzhou Alfa Chemical Co.,Ltd | +8618530059196 | sale04@alfachem.cn | China | 13349 | 58 |
| HANGZHOU CLAP TECHNOLOGY CO.,LTD | 86-571-88216897,88216896 13588875226 | sales@hzclap.com | CHINA | 6313 | 58 |
| Finetech Industry Limited | +86-27-87465837 +8618971612321 | info@finetechnology-ind.com | China | 9640 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354 | support@targetmol.com | United States | 19973 | 58 |
| LEAPCHEM CO., LTD. | +86-852-30606658 | market18@leapchem.com | China | 43348 | 58 |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 | product@acmec-e.com | China | 33350 | 58 |
13220-57-0(TRYPTANTHRIN)Related Search:
1of4