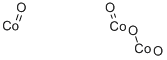Tricobalt tetraoxide
- CAS No.
- 1308-06-1
- Chemical Name:
- Tricobalt tetraoxide
- Synonyms
- cobalt Tetroxide;Co3-O4;COBALT BLACK;Cobalt oxide (Co3O4);COBALT(III) OXIDE BLACK;Cobalt(II;CobaltOxideGr;COBALTIC OXIDE;cobaltosicoxide;CobaltOxidePure
- CBNumber:
- CB5853023
- Molecular Formula:
- Co3O4-2
- Molecular Weight:
- 240.8
- MDL Number:
- MFCD00010939
- MOL File:
- 1308-06-1.mol
- MSDS File:
- SDS
| Melting point | 895 °C (dec.)(lit.) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boiling point | 3800°C | ||||||||||||||
| Density | 6.11 g/mL at 25 °C(lit.) | ||||||||||||||
| vapor pressure | 0Pa at 20℃ | ||||||||||||||
| solubility | insoluble in H2O; soluble in acid solutions, alkaline solutions | ||||||||||||||
| form | powder | ||||||||||||||
| color | Dark Gray | ||||||||||||||
| Specific Gravity | 6.11 | ||||||||||||||
| Water Solubility | Soluble in acids and alkalis. Insoluble in water. | ||||||||||||||
| Crystal Structure | Spinel type | ||||||||||||||
| crystal system | Cube | ||||||||||||||
| Merck | 14,2429 | ||||||||||||||
| Space group | Fd3m | ||||||||||||||
| Lattice constant |
|
||||||||||||||
| Exposure limits | ACGIH: TWA 0.02 mg/m3 | ||||||||||||||
| InChI | InChI=1S/3Co.4O | ||||||||||||||
| InChIKey | LBFUKZWYPLNNJC-UHFFFAOYSA-N | ||||||||||||||
| SMILES | O([Co]=O)[Co]=O.O=[Co] | ||||||||||||||
| CAS DataBase Reference | 1308-06-1(CAS DataBase Reference) | ||||||||||||||
| EWG's Food Scores | 1 | ||||||||||||||
| FDA UNII | USK772NS56 | ||||||||||||||
| NIST Chemistry Reference | Cobalt oxide(1308-06-1) | ||||||||||||||
| EPA Substance Registry System | Cobalt tetraoxide (1308-06-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H334-H350i-H412 | |||||||||
| Precautionary statements | P201-P273-P308+P313 | |||||||||
| Hazard Codes | Xn,T,Xi | |||||||||
| Risk Statements | 36/37/38-40-43-49-50/53-42-52/53 | |||||||||
| Safety Statements | 26-36/37/39-45-36/37-53-61-22 | |||||||||
| RIDADR | 1477 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | GG2500000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 9 | |||||||||
| HS Code | 28220000 | |||||||||
| NFPA 704 |
|
Tricobalt tetraoxide price More Price(30)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 221643 | Cobalt(II,III) oxide powder, <10 μm | 1308-06-1 | 50g | $96.1 | 2024-03-01 | Buy |
| Sigma-Aldrich | 203114 | Cobalt(II,III) oxide ≥99.99% trace metals basis | 1308-06-1 | 5g | $114 | 2023-01-07 | Buy |
| Alfa Aesar | 010695 | Cobalt(II,III) oxide, Puratronic?, 99.9985% (metals basis) | 1308-06-1 | 10g | $137 | 2024-03-01 | Buy |
| Alfa Aesar | 010695 | Cobalt(II,III) oxide, Puratronic?, 99.9985% (metals basis) | 1308-06-1 | 50g | $568 | 2023-06-20 | Buy |
| Strem Chemicals | 27-0480 | Cobalt oxide-molybdenum oxide on alumina (3.5% CoO, 14% MoO3) | 1308-06-1 | 500g | $266 | 2021-12-16 | Buy |
Tricobalt tetraoxide Chemical Properties,Uses,Production
Description
Cobalt Oxide (II, III) is a kind oxide of cobalt. It has applications in enamels, semiconductors, grinding wheels. As an important inorganic material, it can be used in catalysis, superconductors, ceramics and other fields. It can also be used as catalyst and catalyst carrier, as electrode active material, for glass, porcelain colorants and pigments, as chemical industry oxidant and as a catalyst for organic synthesis, in senior goggles and other filter materials, in temperature and gas sensors, in electronic ceramics, in lithium ion battery electrode materials, magnetic materials, electrochromic devices, enamels, solar energy absorbers. Cobalt oxide nanoparticles can readily enter cells, and have the potential to be used for hyperthermic treatment, gene therapy and drug delivery. It is more commonly applied for energy storage such as Lithium-ion battery. It may also be used as a gas sensor in sensing toluene, acetone and some other toxic vapors.
Preparation
Tricobalt tetroxide is obtained when cobalt(II) carbonate, cobalt(II) or cobalt(III) oxide, or cobalt hydroxide oxide, CoO(OH) is heated in air at temperatures above 265°C. The temperature must not exceed 800°C (see decomposition temperature above).
Reactions
Heating above 900°C expels oxygen out of the molecule forming cobalt(II) oxide:
2Co3O4 6CoO + O2
Tricobalt tetroxide absorbs oxygen at lower temperatures, but there is no change in the crystal structure.)
The oxide is reduced to its metal by hydrogen, carbon or carbon monoxide.
Co3O4 + 4H2 3Co + 4H2O
Co3O4 + 4CO 3Co + 4CO2
Sources
https://en.wikipedia.org/wiki/Cobalt(II,III)_oxide
https://www.alfa.com/zh-cn/catalog/040184/
https://en.wikipedia.org/wiki/Cobalt_oxide_nanoparticle
https://pubchem.ncbi.nlm.nih.gov/compound/6432046#section=Top
Chemical Properties
Black fine powder
Physical properties
Black cubic crystal; density 6.11 g/cm3; decomposes above 900°C, losing oxygen; insoluble in water; soluble in acids and alkalis.
Uses
It is used in artificial photosynthesis. It is used as a magnetic semiconductor. It is also employed in supercapacitors and lithium-ion batteries.
Uses
In enamels; in semiconductors; in grinding wheels.
Flammability and Explosibility
Not classified
Tricobalt tetraoxide Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Zhengzhou Alfa Chemical Co.,Ltd | +8618530059196 | sale04@alfachem.cn | China | 13349 | 58 |
| City Chemical LLC | 2039322489 | sales@citychemical.com | United States | 168 | 58 |
| henan kanbei chemical co.,ltd | +undefined-86-1523780-4566 +undefined15237804566 | henankanbeichemical@163.com | China | 511 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-81148696 +86-15536356810 | 1022@dideu.com | China | 3878 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | sales@hbmojin.com | China | 12446 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 | peter@yan-xi.com | China | 5873 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | info@dakenam.com | China | 18628 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21667 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29888 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28180 | 58 |
View Lastest Price from Tricobalt tetraoxide manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-08-19 | Tricobalt tetraoxide
1308-06-1
|
US $0.00-0.00 / KG | 1KG | 99.0% | 10000KG | Shaanxi Dideu Medichem Co. Ltd | |
 |
2024-08-15 | Tricobalt tetraoxide
1308-06-1
|
US $20.00 / kg | 1kg | 99% | 30 tons | Hebei Yanxi Chemical Co., Ltd. | |
 |
2023-08-16 | Tricobalt tetraoxide
1308-06-1
|
US $60.00 / KG | 25KG | 99.99% | 200ton | Hebei Mojin Biotechnology Co., Ltd |
-

- Tricobalt tetraoxide
1308-06-1
- US $0.00-0.00 / KG
- 99.0%
- Shaanxi Dideu Medichem Co. Ltd
-

- Tricobalt tetraoxide
1308-06-1
- US $20.00 / kg
- 99%
- Hebei Yanxi Chemical Co., Ltd.
-

- Tricobalt tetraoxide
1308-06-1
- US $60.00 / KG
- 99.99%
- Hebei Mojin Biotechnology Co., Ltd
1308-06-1(Tricobalt tetraoxide)Related Search:
1of4