5-NITRO BENZOXAZOLO-2-THIONE
- CAS No.
- 22876-21-7
- Chemical Name:
- 5-NITRO BENZOXAZOLO-2-THIONE
- Synonyms
- Ilaprazole Impurity 40;5-NITRO BENZOXAZOLO-2-THIONE;5-Nitrobenzo[d]oxazole-2(3H);5-Nitro-2(3H)-benzoxazolethione;2(3H)-Benzoxazolethione,5-nitro-;5-Nitro-2(3H)-benzodoxazolethione;5-Nitrobenzo[d]oxazole-2(3H)-thione;5-Nitro-3H-1,3-benzoxazole-2-thione;5-Nitro-benzo[d]oxazol-2(3H)-thione;5-Nitrobenzo[d]oxazole-2-thiol
- CBNumber:
- CB8461522
- Molecular Formula:
- C7H4N2O3S
- Molecular Weight:
- 196.18
- MDL Number:
- MFCD00785214
- MOL File:
- 22876-21-7.mol
- MSDS File:
- SDS
| Product description | Number | Pack Size | Price |
| 5-Nitro-1,3-benzoxazole-2-thiol | N497168 | 100mg | $60 |
| 5-Nitrobenzo[d]oxazole-2(3H)-thione | Z4454 | 100mg | $73 |
| 5-Nitrobenzo[d]oxazole-2(3H)-thione | Z4454 | 250mg | $80 |
| 5-Nitro-1,3-benzoxazole-2-thiol 95% | OR965833 | 1g | $111 |
| 5-Nitro-1,3-benzoxazole-2-thiol 95% | OR965833 | 5g | $375 |
| More product size | |||
| Melting point | 244-245 °C |
|---|---|
| Boiling point | 326.7±44.0 °C(Predicted) |
| Density | 1.65±0.1 g/cm3(Predicted) |
| storage temp. | 2-8°C |
| form | solid |
| pka | 8.90±0.20(Predicted) |
| Appearance | Light brown to brown Solid |
| InChI | 1S/C7H4N2O3S/c10-9(11)4-1-2-6-5(3-4)8-7(13)12-6/h1-3H,(H,8,13) |
| InChIKey | FQOGSTGLRLMQOV-UHFFFAOYSA-N |
| SMILES | SC1=NC2=CC([N+]([O-])=O)=CC=C2O1 |
| UNSPSC Code | 12352105 |
| NACRES | NA.21 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P261-P305+P351+P338 |
| WGK Germany | WGK 3 |
| HS Code | 2934999090 |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Acute Tox. 4 Oral |
5-NITRO BENZOXAZOLO-2-THIONE price
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| TRC | N497168 | 5-Nitro-1,3-benzoxazole-2-thiol | 22876-21-7 | 100mg | $60 | 2021-12-16 | Buy |
| AK Scientific | Z4454 | 5-Nitrobenzo[d]oxazole-2(3H)-thione | 22876-21-7 | 100mg | $73 | 2021-12-16 | Buy |
| AK Scientific | Z4454 | 5-Nitrobenzo[d]oxazole-2(3H)-thione | 22876-21-7 | 250mg | $80 | 2021-12-16 | Buy |
| Apolloscientific | OR965833 | 5-Nitro-1,3-benzoxazole-2-thiol 95% | 22876-21-7 | 1g | $111 | 2021-12-16 | Buy |
| Apolloscientific | OR965833 | 5-Nitro-1,3-benzoxazole-2-thiol 95% | 22876-21-7 | 5g | $375 | 2021-12-16 | Buy |
5-NITRO BENZOXAZOLO-2-THIONE Chemical Properties,Uses,Production
Uses
5-Nitro-1,3-benzoxazole-2-thiol is used in preparation of benzazole-substituted benzo-fused oxaheterocycle carboxamides as NOX4 inhibitors for treatment of diseases involving impaired reactive oxygen species generation such as fibrosis.
Synthesis
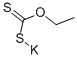
140-89-6
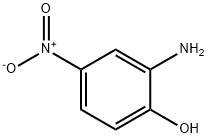
99-57-0
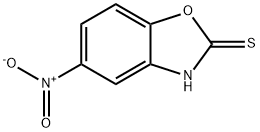
22876-21-7
GENERAL METHOD: Potassium ethylxanthate (1872.4 mg, 11.7 mmol, 2 equiv) was added to a solution of 2-amino-4-nitrophenol (5.8 mmol, 1 equiv) in 25 mL of anhydrous ethanol. The reaction mixture was heated to reflux for 4 hours. Upon completion of the reaction, it was cooled to room temperature and concentrated to dryness under reduced pressure. The residue was dissolved in water and acidified with acetic acid to pH 5. The precipitate was collected by filtration, washed with water and dried for 48 h to give 5-nitrobenzoxazole-2-thiol, which can be used without further purification. Iodomethane (1.2 eq.) and anhydrous potassium carbonate (1 eq.) were added to a solution of 5-nitrobenzoxazole-2-thiol (1 eq.) in acetonitrile (15 mL). The reaction mixture was stirred for 4 hours at room temperature. Upon completion of the reaction, the reaction was quenched by the addition of a drop of water and the solvent was removed under reduced pressure. The residue was suspended in water and extracted three times with ethyl acetate. The organic phases were combined, washed with brine (110 mL) and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure to give 2-(methylthio)-5-nitrobenzoxazole, which can be used without further purification. 2-(Methylthio)-5-nitrobenzoxazole (1 eq.) was mixed with the appropriate amine (3 eq.) in acetonitrile and heated in a microwave reactor at 120 °C for 20-30 min. Upon completion of the reaction, the solvent was removed under reduced pressure, the residue was dissolved in ethyl acetate, washed sequentially with water (2 x 10 mL), saturated sodium bicarbonate solution (used to neutralize hydrobromic acid only if bromine-substituted reactants were used), brine (10 mL) and dried over anhydrous magnesium sulfate. The solvent was removed under reduced pressure to give the crude product. Purification by column chromatography using a gradient elution of methanol/ethyl acetate (0-20%) or hexane/ethyl acetate gave the pure product. The aromatic nitro compound (1 mmol) was dissolved in ethanol (8 mL) in a heated flask. Add 10% palladium/carbon (0.3 times the mass of the reduced compound for the catalyst). The reaction flask was sealed, filled with hydrogen and evacuated twice. The reaction mixture was stirred on a Parr shaker at 60 psi hydrogen pressure for 12 h. The progress of the reaction was monitored by thin layer chromatography. Upon completion of the reaction, the aromatic amine was obtained by filtration through diatomaceous earth and the solvent was removed under reduced pressure. The aromatic amine (1 mmol, 1 eq.) was mixed with 4,7-dichloroquinoline (1.1 eq.) in acetonitrile (25 mL). It was acidified by adding 3-5 drops of hydrochloric acid and stirred at reflux for 24 hours. Upon completion of the reaction, the solvent was removed under reduced pressure and the residue was dissolved in ethyl acetate (or 20% methanol, against a methanolic solution of the toluene compound) and washed sequentially with saturated sodium bicarbonate solution (3 x 20 mL) and brine (110 mL). The organic phases were combined, dried over anhydrous magnesium sulfate, adsorbed on silica gel (5 g), and subjected to silica gel column chromatography for separation, resulting in 2-mercapto-5-nitrobenzoxazole.
References
[1] Bioorganic and Medicinal Chemistry Letters, 2012, vol. 22, # 15, p. 5046 - 5050
[2] Bioorganic and Medicinal Chemistry, 2015, vol. 23, # 17, p. 5419 - 5432
[3] Patent: WO2007/91106, 2007, A2. Location in patent: Page/Page column 133
[4] Patent: WO2006/66174, 2006, A1. Location in patent: Page/Page column 121
[5] Patent: EP1219622, 2002, A2. Location in patent: Page 23
5-NITRO BENZOXAZOLO-2-THIONE Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 33024 | 60 |
| Aladdin Scientific | tp@aladdinsci.com | United States | 52923 | 58 | |
| DAYANG CHEM (HANGZHOU) CO.,LTD | +86-88938639 +86-17705817739 | info@dycnchem.com | China | 53802 | 58 |
| Amadis Chemical Company Limited | 571-89925085 | sales@amadischem.com | China | 131957 | 58 |
| Nanjing Chemlin Chemical Co., Ltd | 025-83697070 13770331960 | info@chemlin.com.cn | China | 16059 | 64 |
| S.Z. PhyStandard Bio-Tech. Co., Ltd. | 0755-4000505016 13380397412 | 3001272453@qq.com | China | 5157 | 50 |
| Nanjing Norris-Pharm Technology Co., Ltd | 18652989687 | sales@norris-pharm.com | China | 8878 | 55 |
| Shanghai Jian Chao Chemical Technology Co., Ltd. | 150-2103-5486 18017383231 | 983544897@qq.com | China | 9341 | 55 |
| Beijing FYF Chemicals Co., Ltd | 010-57903446 15869909019 | 168931144@qq.com | China | 2873 | 58 |
| Shanghai Kewel Chemical Co., Ltd. | 021-64609169 18901607656 | greensnown@163.com | China | 9906 | 50 |





