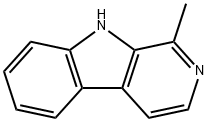
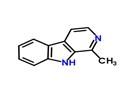
HARMANE
- CAS No.
- 486-84-0
- Chemical Name:
- HARMANE
- Synonyms
- HARMAN;1-METHYL-9H-PYRIDO[3,4-B]INDOLE;1-METHYL-PYRIDO[3,4-B]INDOLE;Aribin;ARIBINE;HARMANE;Indoter;Loturine;NSC 54439;Locuturin
- CBNumber:
- CB9698060
- Molecular Formula:
- C12H10N2
- Molecular Weight:
- 182.22
- MDL Number:
- MFCD00004957
- MOL File:
- 486-84-0.mol
- MSDS File:
- SDS
| Melting point | 235-238 °C(lit.) |
|---|---|
| Boiling point | 305.62°C (rough estimate) |
| Density | 1.1485 (rough estimate) |
| refractive index | 1.6266 (estimate) |
| storage temp. | Store at RT |
| solubility | methanol: soluble50mg/ml |
| pka | 7.37, 14.6(at 25℃) |
| form | Solid |
| color | White to Dark Brown |
| Water Solubility | 1523g/L(20 ºC) |
| Merck | 13,4630 |
| BRN | 143898 |
| LogP | 3.100 |
| CAS DataBase Reference | 486-84-0(CAS DataBase Reference) |
| FDA UNII | 82D6J0535P |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H302-H315-H319-H332-H335 |
| Precautionary statements | P261-P280-P305+P351+P338 |
| Hazard Codes | Xn,Xi |
| Risk Statements | 20/21-36/37/38 |
| Safety Statements | 22-24/25-36-26 |
| RIDADR | 1544 |
| WGK Germany | 3 |
| RTECS | UV0280000 |
| HazardClass | 6.1(b) |
| PackingGroup | III |
| Toxicity | LD50 i.p. in mice: 50 mg/kg (Sigg) |
HARMANE price More Price(19)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 103276 | Harmane 98% | 486-84-0 | 1g | $246 | 2024-03-01 | Buy |
| Cayman Chemical | 29613 | Harmane | 486-84-0 | 250mg | $56 | 2021-12-16 | Buy |
| Cayman Chemical | 29613 | Harmane | 486-84-0 | 500mg | $91 | 2021-12-16 | Buy |
| Cayman Chemical | 29613 | Harmane | 486-84-0 | 100mg | $50 | 2024-03-01 | Buy |
| Cayman Chemical | 29613 | Harmane | 486-84-0 | 1g | $226 | 2024-03-01 | Buy |
HARMANE Chemical Properties,Uses,Production
Chemical Properties
Off-White Solid
Uses
- Harmane was used in trace level determination of harmane by planar chromatography coupled with (tandem) mass spectrometry.
- It was used to study interactions of norharman and harman with DNA.
- It may be used as matrix for analysis of cyclodextrins and for sulfated oligosaccharides in combination with DHB as co-matrix.
Uses
Harman alkaloid like harmane, harmine, harmalol, harmaline obtained from Banisteriopsis caapi L. showed cytotoxicity, antimicrobial activity against Staphylococcus aureus, Escherichia coli, Proteus vulgaris and Candida albicans.
Definition
ChEBI: An indole alkaloid fundamental parent with a structure of 9H-beta-carboline carrying a methyl substituent at C-1. It has been isolated from the bark of Sickingia rubra, Symplocus racemosa, P ssiflora incarnata, Peganum harmala, Banisteriopsis caapi and Tribulus terrestris, as well as from tobacco smoke. It is a specific, reversible inhibitor of monoamine oxidase A.
Synthesis Reference(s)
The Journal of Organic Chemistry, 37, p. 1429, 1972 DOI: 10.1021/jo00974a030
Tetrahedron, 49, p. 3325, 1993 DOI: 10.1016/S0040-4020(01)90161-9
General Description
- Harmane is a potent tremor-producing β-carboline alkaloid and neurotoxin.
- It is major representative of heterocyclic aromatic amines, a group of mutagenic and carcinogenic substances which are formed in meat from the precursors creatine, creatinine, amino acids and sugars during the heating at high temperatures.
- Blood harmane concentration is elevated in essential tremor, late-life neurological disease.
Biological Activity
Proposed as the endogenous ligand for imidazoline binding sites. Binds to I 1 -sites in rat kidney with an IC 50 of 31 nM, and I 2 -sites with a K i of 49 nM. In vivo, produces a dose-dependent hypotension that is reversed by efaroxan (2-(2-Ethyl-2,3-dihydro-2-benzofuranyl)-4,5-dihydro-1H-imidazole hydrochloride ). Also a potent inhibitor of monoamine oxidases A and B (I 50 values are 0.5 and 5 μ M respectively).
Biochem/physiol Actions
I1 imidazoline binding site agonist.
storage
Room temperature
HARMANE Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 | zheyansh@163.com | CHINA | 3620 | 58 |
| career henan chemical co | +86-0371-86658258 15093356674; | factory@coreychem.com | China | 29826 | 58 |
| Zhengzhou Alfa Chemical Co.,Ltd | +8618530059196 | sale04@alfachem.cn | China | 12468 | 58 |
| Hubei Ipure Biology Co., Ltd | +8613367258412 | ada@ipurechemical.com | China | 10326 | 58 |
| HONG KONG IPURE BIOLOGY CO.,LIMITED | 86 18062405514 18062405514 | ada@ipurechemical.com | CHINA | 3465 | 58 |
| LEAP CHEM CO., LTD. | +86-852-30606658 | market18@leapchem.com | China | 24738 | 58 |
| TargetMol Chemicals Inc. | +1-781-999-5354 | support@targetmol.com | United States | 19973 | 58 |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 | product@acmec-e.com | China | 33349 | 58 |
| Aladdin Scientific | +1-833-552-7181 | sales@aladdinsci.com | United States | 57511 | 58 |
| Amadis Chemical Company Limited | 571-89925085 | sales@amadischem.com | China | 131981 | 58 |