Genipin
- CAS No.
- 6902-77-8
- Chemical Name:
- Genipin
- Synonyms
- enipin;GENIPIN;Jingping;Genipin>GENIPIN(SH);GENIPIN(RG);(+)-Genipin;Genipin(AS);Genipin 95%;Genipin, >=98%
- CBNumber:
- CB9767199
- Molecular Formula:
- C11H14O5
- Molecular Weight:
- 226.23
- MDL Number:
- MFCD00888600
- MOL File:
- 6902-77-8.mol
- MSDS File:
- SDS
- TDS File:
- TDS
| Product description | Number | Pack Size | Price |
| Genipin ≥98% (HPLC), powder | G4796 | 25mg | $148 |
| Genipin phyproof? Reference Substance | PHL80458 | 10MG | $532 |
| Genipin ≥98% (HPLC), powder | G4796 | 125mg | $586 |
| Genipin >97.0%(GC) | G0458 | 25mg | $57 |
| Genipin >97.0%(GC) | G0458 | 100mg | $174 |
| More product size | |||
| Melting point | 118.0 to 123.0 °C |
|---|---|
| Boiling point | 287.83°C (rough estimate) |
| Density | 1.1230 (rough estimate) |
| refractive index | 1.4720 (estimate) |
| storage temp. | Inert atmosphere,Room Temperature |
| solubility | DMSO: ≥25mg/mL |
| form | powder |
| pka | 12.06±0.60(Predicted) |
| color | White to Almost white |
| optical activity | 117.7° (C=0.01 g/ml, MEOH) |
| λmax | 240nm(MeOH)(lit.) |
| InChI | InChI=1S/C11H14O5/c1-15-10(13)8-5-16-11(14)9-6(4-12)2-3-7(8)9/h2,5,7,9,11-12,14H,3-4H2,1H3/t7-,9-,11-/m1/s1 |
| InChIKey | AZKVWQKMDGGDSV-BCMRRPTOSA-N |
| SMILES | [C@@H]1(O)OC=C(C(OC)=O)[C@@]2([H])CC=C(CO)[C@@]12[H] |
| CAS DataBase Reference | 6902-77-8(CAS DataBase Reference) |
| FDA UNII | A3V2NE52YG |
| UNSPSC Code | 41116107 |
| NACRES | NA.77 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301 | |||||||||
| Precautionary statements | P301+P310+P330 | |||||||||
| PPE | dust mask type N95 (US), Eyeshields, Faceshields, Gloves | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 22 | |||||||||
| Safety Statements | 3/14-36/37/39 | |||||||||
| RIDADR | UN 2811 6.1/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | GY5828000 | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 29329990 | |||||||||
| Storage Class | 6.1C - Combustible acute toxic Cat.3 toxic compounds or compounds which causing chronic effects |
|||||||||
| Hazard Classifications | Acute Tox. 3 Oral | |||||||||
| NFPA 704 |
|
Genipin price More Price(67)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | G4796 | Genipin ≥98% (HPLC), powder | 6902-77-8 | 25mg | $148 | 2026-04-30 | Buy |
| Sigma-Aldrich | PHL80458 | Genipin phyproof? Reference Substance | 6902-77-8 | 10MG | $532 | 2026-04-30 | Buy |
| Sigma-Aldrich | G4796 | Genipin ≥98% (HPLC), powder | 6902-77-8 | 125mg | $586 | 2026-04-30 | Buy |
| TCI Chemical | G0458 | Genipin >97.0%(GC) | 6902-77-8 | 25mg | $57 | 2026-04-30 | Buy |
| TCI Chemical | G0458 | Genipin >97.0%(GC) | 6902-77-8 | 100mg | $174 | 2026-04-30 | Buy |
Genipin Chemical Properties,Uses,Production
Chemical Properties
It is a white crystalline powder that is soluble in organic solvents such as methanol, ethanol, and DMSO. It is derived from the fruit of Gardenia jasminoides Ellis.
Uses
Genipin has been used:
- in chemosensitivity assay
- in the preparation of recombinant human (rh)-odontogenic ameloblast-associated protein (ODAM) -impregnated collagen gel and in vitro mineralization assay
- in genipin gel preparation
Uses
Genipin is an active aglycone derived from geniposide, an iridoid glycoside found in the fruit of Gardenia jasminoides Ellis. It has been used in traditional Chinese medicine and is a hydrolytic product of geniposide. Genipin is also known for its inhibitory effect on uncoupling protein 2 (UCP2).
Definition
ChEBI: Genipin is an iridoid monoterpenoid. It has a role as an uncoupling protein inhibitor, a hepatotoxic agent, an apoptosis inhibitor, an antioxidant, an anti-inflammatory agent and a cross-linking reagent.
General Description
Genipin is a natural cross linking agent, which is extracted from gardenia fruit. It prevents lipid peroxidation and production of nitric oxide. Genipin protects the hippocampal neurons from the toxicity of Alzheimer′s amyloid β protein. It has anti-inflammatory and anti-angiogenesis effects. Genipin is involved in drug delivery system.
Biochem/physiol Actions
Genipin stimulates insulin secretion in UCP2-dependent manner (Uncoupling protein 2). Genipin is a protein, collagen, gelatin, and chitosan cross-linker.
Synthesis
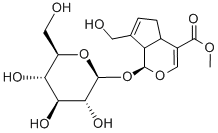
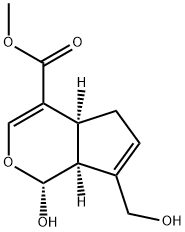
24512-63-8
![Cyclopenta[c]pyran-4-carboxylic acid, 1,4a,5,7a-tetrahydro-1-hydroxy-7-(hydroxymethyl)-, methyl ester, [1S-(1α,4aβ,7aβ)]- (9CI)](/CAS/20210305/GIF/70094-79-0.gif)
70094-79-0
Synthesis of the compound (CAS:70094-79-0) from (1S,4aS,7aS)-methyl 7-(hydroxymethyl)-1-(((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-yl)oxy)-1,4a,5,7a-tetrahydropenta[c]pyran-4-carboxylate The general steps of the synthesis were as follows: 1. 1 g of gardenia glycoside was dissolved in 30 mL of dichloromethane/water (1:1, v/v) and 100 mg of β-glucosidase was added. The reaction mixture was stirred vigorously at 37°C for 24 hours. After completion of the reaction, it was extracted three times with dichloromethane. The organic layers were combined, dried over anhydrous sodium sulfate and concentrated under reduced pressure to give 487 mg of genipin in 84% yield as a white powder. 2. 50 mg of kyonipin (0.22 mmol) was dissolved in dichloromethane and 53 mg of di-tert-butyl dicarbonate (Boc2O, 0.24 mmol) and 0.15 mL of triethylamine (Et3N, 1.11 mmol) were added sequentially. The reaction mixture was stirred for 3 hours. After completion of the reaction, 1N hydrochloric acid was added and the organic layer was separated, washed with saturated brine, dried over anhydrous sodium sulfate and concentrated under reduced pressure. The residue was purified by silica gel column chromatography (eluent: hexane solution of 10% ethyl acetate) to give 51 mg of compound (i) in 71% yield as a colorless oil. 3. Compound (i) (1 eq.) was dissolved in anhydrous dichloromethane and 1.1 eq. of N-Boc-amino acid, 1.1 eq. of 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDC-HCl) and 0.1 eq. of 4-dimethylaminopyridine (DMAP) were added sequentially. The reaction mixture was stirred at room temperature until the ingredients were completely consumed. After completion of the reaction, it was diluted with dichloromethane and washed three times with saturated saline. The organic layer was dried with anhydrous sodium sulfate, concentrated under reduced pressure and the residue was separated by gel chromatography to give compound (ii) in 99% yield. 4. Compound (ii) (1 eq.) was dissolved in a mixture of 25 eq. of trifluoroacetic acid (TFA) and dichloromethane (CH2Cl2/TFA, 10:1, v/v), and the reaction was stirred for 2 hours. After completion of the reaction, trifluoroacetic acid was removed by concentration under reduced pressure. The residue was purified by silica gel column chromatography (eluent: 5% dichloromethane solution in methanol) to give compound 1 in 87% yield.
References
[1] Patent: US2017/298049, 2017, A1. Location in patent: Paragraph 0095; 0096; 0097; 0098
[2] Molecules, 2011, vol. 16, # 5, p. 4295 - 4304
[3] Journal of Agricultural and Food Chemistry, 2001, vol. 49, # 1, p. 430 - 432
[4] Heterocycles, 1991, vol. 32, # 8, p. 1451 - 1454
[5] Natural Product Research, 2012, vol. 26, # 8, p. 765 - 769
Genipin Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 | deasea125996@gmail.com | China | 2472 | 58 |
| Jinan Ruitong Biotech Co., Ltd. | +86-16615778970 +86-16615778970 | info@ritonbio.com | China | 511 | 58 |
| Hebei Chuanghai Biotechnology Co., Ltd | +8615350571055 | Sibel@chuanghaibio.com | China | 8738 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +8617392709771 | 1097@dideu.com | China | 3996 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 | info@fdachem.com | China | 20124 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618092446649 | sarah@tnjone.com | China | 1143 | 58 |
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 | admin@zlchemi.com | China | 3692 | 58 |
| Capot Chemical Co.,Ltd. | +86-(0)57185586718; +8613336195806 | sales@capot.com | China | 29640 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21590 | 55 |
| Shanghai Zheyan Biotech Co., Ltd. | 18017610038 | zheyansh@163.com | CHINA | 3619 | 58 |
Related articles
- Pharmacology research of Genipin
- Genipin is an important cross-linking agent due to its tunable properties and versatile applications such as food,medical, and....
- Oct 15,2025
View Lastest Price from Genipin manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2026-05-04 | 1,4a,5,7a-Tetrahydro-1-hydroxy-7-(hydroxymethyl)-cyclopenta(c)pyran-4-carboxylic acid methyl ester
6902-77-8
|
0.99 | RongNa Biotechnology Co.,Ltd | ||||
 |
2026-04-30 | Genipin
6902-77-8
|
US $0.00 / g | 1g | ≥98.0% | 10kg/month | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2026-04-22 | Genipin
6902-77-8
|
US $50.00 / mg | 99.09% | 10g | TargetMol Chemicals Inc. |
-

- 1,4a,5,7a-Tetrahydro-1-hydroxy-7-(hydroxymethyl)-cyclopenta(c)pyran-4-carboxylic acid methyl ester
6902-77-8
- 0.99
- RongNa Biotechnology Co.,Ltd
-

- Genipin
6902-77-8
- US $0.00 / g
- ≥98.0%
- WUHAN FORTUNA CHEMICAL CO., LTD
-

- Genipin
6902-77-8
- US $50.00 / mg
- 99.09%
- TargetMol Chemicals Inc.
6902-77-8(Genipin)Related Search:
1of4