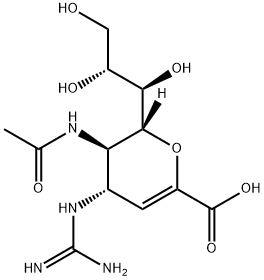
ZANAMIVIR HYDRATE
|
|
ZANAMIVIR HYDRATE 속성
- 녹는점
- 2560C (dec.)
- 알파
- D20 +40.9° (c = 0.9 in water)
- 밀도
- 1.75±0.1 g/cm3(Predicted)
- 저장 조건
- 2-8°C
- 용해도
- H2O: 가용성10mg/mL, 투명
- 물리적 상태
- 가루
- 산도 계수 (pKa)
- 3.82±0.70(Predicted)
- 색상
- 흰색에서 베이지색
- optical activity
- [α]/D +30 to +40°, c = 1 in H2O
- 수용성
- Soluble to 5 mM in water
- 안정성
- 흡습성
- InChIKey
- ARAIBEBZBOPLMB-UFGQHTETSA-N
- CAS 데이터베이스
- 139110-80-8
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xn | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 22-36/37/38 | ||
| 안전지침서 | 26 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | RA9451000 | ||
| HS 번호 | 2932999000 | ||
| 유해 물질 데이터 | 139110-80-8(Hazardous Substances Data) |
ZANAMIVIR HYDRATE C화학적 특성, 용도, 생산
개요
Zanamivir was launched as Relenza in Australia for treatment of human influenza A and B virus infections. Zanamivir (4-guanidino-Neu5Ac2en) can be obtained by several similar ways, for instance in seven step synthesis starting from N-acetyI-D-neuraminic acid. Mechanistically, Zanamivir is a potent and specific inhibitor of neuraminidase (or sialidase), a key viral surface glycohydrolase essential for viral replication and disease progression by catalyzing the cleavage of terminal sialic acid residues from the glycoprotein. The in vitro activity of Zanamivir against a wide variety of influenza A and B strains was demonstrated in different model systems; its activity against clinically relevant isolates of influenza virus, with IC50 values ranging from 0.005 to 15 pM was superior to those of amantadine and rimantadine.화학적 성질
Colorless Crystalline Solid용도
ZANAMIVIR HYDRATE is a sialic acid analog that inhibits neuraminidase release of newly replicated influenza virus particles. It has been shown to selectively inhibit the growth of influenza A and B viruses in plaque reduction assays with IC50 values ranging from 5 to 14 nM and to directly inhibit influenza A and B virus neuraminidases with IC50 values ranging from 0.6 to 7.9 nM in vitro. The efficacy and tolerabilbity of zanamivir has also been established in clinical trial.[Cayman Chemical]Indications
Zanamivir (Relenza) is a neuraminidase inhibitor with activity against influenza A and B strains. Like oseltamivir, zanamivir is a reversible competitive antagonist of viral neuraminidase. It inhibits the release of progeny virus, causes viral aggregation at the cell surface, and impairs viral movement through respiratory secretions. Resistant variants with hemagglutinin and/or neuraminidase mutations have been produced in vivo; however, clinical resistance to zanamivir is quite rare at present.원료
Resistance is presently uncommon, including strains resistant to oseltamivir. In clinical trials the frequency was no more than 1% of exposed patients.일반 설명
Zanamivir is identical to 2-deoxy-2,3-dehydro-N-acetylneuraminic acid except that itpossesses a guanidino group at position 4 instead of a hydroxylgroup. At positions 119 and 227 of the receptor site,there exist glutamic acid residues. Zanamivir has beenshown to form a salt bridge with the guanidine and Glu-119and a charge transfer interaction with Glu-227. These interactionsincrease the interaction strength with the enzymeand create an excellent competitive inhibitor and an effectiveantiviral agent for influenza types A and B.Human studies have shown that zanamivir is effectivewhen administered before or after exposure to the influenzavirus. If administered before exposure to the virus, the drugreduced viral propagation, infectivity, and disease symptoms.If administered after exposure, the drug reduces propagation,viral titer, and illness. Zanamivir is marketed as a dry powderfor oral inhalation. It is used in adolescents and adults who have been exposed and are symptomatic for not more than 2days. Zanamivir is also indicated for prophylactically treatingfamily members of a person who has developed influenza.
Pharmaceutical Applications
A synthetic neuraminidase inhibitor formulated for administration by inhalation.Pharmacology
Zanamivir is generally well tolerated. Bronchospasm and impaired lung function have been reported in some patients taking this medication, but many of these individuals had serious underlying pulmonary disease. Zanamivir should be discontinued if an individual develops bronchospasm or breathing difficulties; treatment and hospitalization may be required. Allergic reactions, including angioedema, have been rarely reported. The efficacy of zanamivir depends upon the proper use of the inhaler device.Pharmacokinetics
Oral bioavailability is poor. After inhalation local respiratory mucosal concentrations greatly exceed those that are inhibitory for influenza A and B replication. The median concentrations in the sputum exceed 1 mg/L 6 h after inhalation and remain detectable for 24 h.Clinical Use
Treatment of influenza A and B infections in patients over 7 years of age, and prophylaxis of patients ??5 years of age부작용
Most adverse effects are related to the respiratory tree. These include rhinorrhea and, rarely, bronchospasm. Nausea and vomiting have been reported at low incidence.신진 대사
Zanamirvir is effective when administered via the nasal, intraperitoneal, and intravenous (IV) routes, but it is inactive when given orally. Animal studies have shown 68% recovery of the drug in the urine following intraperitoneal administration, 43% urinary recovery following nasal administration, and only 3% urinary recovery following oral administration. Human data gave results similar to those obtained in animal models. Human efficacy studies with nasal drops or sprays demonstrated that the drug was effective when administered before and after exposure to influenza A or B virus. When given before viral inoculation, the drug reduced viral shedding, infection, and symptoms. When administered beginning at either 26 or 32 hours after inoculation, there was a reduction in shedding, viral titer, and fever.ZANAMIVIR HYDRATE 준비 용품 및 원자재
원자재
브롬화 시안
히드라진
메틸아민
아세트산 나트륨
149398-58-3
Carbamimidothioic acid, N-acetyl-, methyl ester
1H-Pyrazole-3-carboximidamide Hydrochloride
D-glycero-D-galacto-Non-2-enonic acid, 5-(acetylamino)-4-[(aminoiminomethyl)amino]-2,6-anhydro-3,4,5-trideoxy-, methyl ester, 7,8,9-triacetate
4-amino-2-deoxy-2,3-didehydro-N-acetylneuraminic acid
Zanamivir
1H-피라졸-1-카르복사미딘 하이드로클로라이드
methyl (3aR,4R,7aR)-2-methyl-4-(1S,2R,3-triacetoxypropyl)-3a,7a-dihydro-4H-pyrano[3,4-d]oxazole-6-carboxylate
AMinoiMinoMethanesulfonic Acid
준비 용품
ZANAMIVIR HYDRATE 공급 업체
글로벌( 272)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 |
info@dakenam.com | China | 15928 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 |
fandachem@gmail.com | China | 9348 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32480 | 60 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Zhejiang ZETian Fine Chemicals Co. LTD | 18957127338 |
stella@zetchem.com | China | 2141 | 58 |
| Biochempartner | 0086-13720134139 |
candy@biochempartner.com | CHINA | 967 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| BOC Sciences | +1-631-485-4226 |
inquiry@bocsci.com | United States | 19553 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 |
sales@chemdad.com | China | 39916 | 58 |
ZANAMIVIR HYDRATE 관련 검색:
리바비린 오셀타미비르 3,4-다이하이드로피란
4-N-tert-Butyloxycarbonyl ZanaMivir AMine
methyl (3aR,4R,7aR)-2-methyl-4-(1S,2R,3-triacetoxypropyl)-3a,7a-dihydro-4H-pyrano[3,4-d]oxazole-6-carboxylate
Ethambutol
Emtricitabine
10,11-Dihydro-alpha-methyl-10-oxo-dibenzo[b,f]thiepin-2-acetic acid
Zalcitabine
Zamicastat
ZanaMivir
1H-Pyrazole-1-carboxamidine hydrochloride
D-GLYCERO-D-GALACTO-NON-2-ENONIC ACID
Zanamivir
4-amino-2-deoxy-2,3-didehydro-N-acetylneuraminic acid
N-Cyano Zanamivir Amine Triacetate Methyl Ester
Zanamivir Azide Methyl Ester
Amantadine