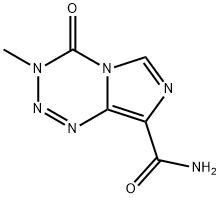
Temozolomide
|
|
Temozolomide 속성
- 녹는점
- 212°C dec.
- 끓는 점
- 526.6±42.0 °C(Predicted)
- 밀도
- 1.97±0.1 g/cm3(Predicted)
- 저장 조건
- 2-8°C
- 용해도
- DMSO: 용해성10mg/mL, 투명
- 산도 계수 (pKa)
- 14.77±0.20(Predicted)
- 물리적 상태
- 가루
- 색상
- 흰색에서 밝은 갈색까지
- Merck
- 14,9139
- 안정성
- 공급된 상태로 2년 동안 안정적입니다. DMSO의 솔루션은 -20°에서 최대 3개월 동안 보관할 수 있습니다.
- InChIKey
- BPEGJWRSRHCHSN-UHFFFAOYSA-N
- CAS 데이터베이스
- 85622-93-1(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 45-46-60-61-22-36/37/38 | ||
| 안전지침서 | 24/25-45-36/37-26-53-36 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | NJ5927050 | ||
| HS 번호 | 29339900 |
Temozolomide C화학적 특성, 용도, 생산
개요
Temozolomide was launched for the first time in the UK for the treatment of patients with glioblastoma multiforme showing recurrence or progression after standard therapy. It can be considered as a cyclic variant of highly reactive triazenes producing a cascade of ionic or radical antitumoral species. Temozolamide is a 3-methyl analog of mitozolomide and was shown to be converted to cytotoxic triazene MCTIC. It can be prepared in 2 steps from 5- aminoimidazole-4-carboxamide by diazotization then cyclization with methylisocyanate. Mechanistically, the depletion of O6-alkylguanine-DNA alkyltransferase (OGAT) in cells or tumors was shown to be correlated with the cytotoxicity of temozolomide which is a potent inhibitor of this enzyme involved in DNA repair activity.Further approval applications for the treatment of other malignant gliomas such as relapsed anaplastic astrocytoma, advanced metastatic melanoma were submitted.
화학적 성질
Off-white to light-pink crystalline solid, crystallized from dichloromethane, melting point 212℃ (decomposition). Maximum UV absorption (in 95% ethanol): 327nm.용도
Temozolomide is an antineoplastic drug and an Imidazotetrazine alkylating agent. It induces apoptosis and causes cell cycle arrest at the G2/M checkpoint. It rapidly degrades in the body, producing the active metabolite MTIC, which exerts an anti-tumor effect. Moreover, temozolomide has shown effectiveness against paclitaxel-resistant tumors and has been utilized in the study of drug resistance mechanisms in glioblastoma cell lines.제조 방법
The synthesis of temozolomide involves several steps. First, 5-Amino-4-imidazolecarboxamide is diazotized using nitrite. Then, the diazotized compound is reacted with methyl isocyanate in dichloromethane. This reaction leads to the cyclization of the compound, ultimately resulting in the formation of temozolomide.정의
ChEBI: An imidazotetrazine that is 3,4-dihydroimidazo[5,1-d][1,2,3,5]tetrazine which is substituted at positions 3, 4, and 8 by methyl, oxo, and carboxamide groups, respectively. A prodrug for MTIC (5-(3-methyltriaz-1-en-1-yl)-1H-i idazole-4-carboxamide, formed by spontaneous hydrolysis of temozolomide in the body), it is used as an oral alkylating agent for the treatment of newly diagnosed malignant glioblastoma multiforme (concomitantly with radiotherapy) and malignant melanoma.일반 설명
Temozolomide can be called as an oral alkylating agent and antitumour drug, which may show potent activity in the treatment of recurrent gliomas or glioblastoma, a kind of tumour that affects the brain or spine.생물학적 활성
DNA methylating, chemotherapeutic agent. Displays antitumor activity against a board spectrum of tumors, including leukemias, lymphomas and solid tumors (IC 50 = 5.0 μ M for cytotoxicity against mouse TLX5 lymphoma cells).Clinical Use
This imidazolotetrazine derivative is administered orally in capsule form for the treatment of glioblastoma multiforme or in patients with anaplastic astrocytoma who have not responded to procarbazine or the nitrosoureas.신진 대사
Oral absorption is rapid and complete. The CYP450 enzymes are not extensively involved in temozolomide metabolism, and less than 6% of the drug is excreted unchanged in the urine. Women clear the drug less effectively than men and have a higher incidence of severe neutropenia and thrombocytopenia in the initial therapy cycle. Food decreases temozolomide absorption, and myelosuppression is the most significant adverse effect.Temozolomide 준비 용품 및 원자재
원자재
5-Amino-4-imidazolecarboxamide
메틸 이소시아네이트
NITROUS ACID
5-(Amino-1-(N-methyl Carbamoyl)
4-Amino-5-imidazolecarboxamide hydrochloride
4-diazo-4H-imidazole-5-carboxamide
4-Nitrophenyl chloroformate
1,5-dihydro-4H-imidazo[4,5-d]-1,2,3-triazin-4-one
준비 용품
Temozolomide 공급 업체
글로벌( 591)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Beijing Mesochem Technology Co.,Ltd | +8613651027935 |
rachel@mesochem.com | China | 191 | 58 |
| Shanghai Huici Pharmaceutical Technology Co., LTD | +8618917134367 |
lixing@pharmahuici.com | China | 133 | 58 |
| Shanghai Huici Pharmaceutical Technology Co., LTD | +8618917134367 |
lixing@pharmahuici.com | China | 133 | 58 |
| Hebei Yanxi Chemical Co., Ltd. | +8617531190177 |
peter@yan-xi.com | China | 5993 | 58 |
| Henan Fengda Chemical Co., Ltd | +86-371-86557731 +86-13613820652 |
info@fdachem.com | China | 7845 | 58 |
| Capot Chemical Co.,Ltd. | 571-85586718 +8613336195806 |
sales@capotchem.com | China | 29797 | 60 |
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 |
sales01@cooperate-pharm.com | CHINA | 1811 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| Nanjing ChemLin Chemical Industry Co., Ltd. | 025-83697070 |
product@chemlin.com.cn | CHINA | 3012 | 60 |
| Nanjing Finetech Chemical Co., Ltd. | 025-85710122 17714198479 |
sales@fine-chemtech.com | CHINA | 885 | 55 |