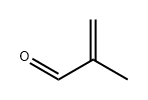
메타크롤레인 C화학적 특성, 용도, 생산
화학적 성질
colourless to light yellow liquid
용도
Methacrolein was used to study the effect of parts per billion levels of limonene oxidation products and the terpene oxidation product, methacrolein on human eye blink frequency. It was also used in determination of rate constants and the products of the reactions between atomic chlorine and acrolein, methacrolein and methyl vinyl ketone.
일반 설명
A colorless liquid. Flash point 35°F. Less dense than water. May be toxic by ingestion, inhalation and skin absorption. Severely irritating to skin and eyes. Used to make plastics.
공기와 물의 반응
Highly flammable. Soluble in water.
반응 프로필
Methacrolein is an aldehyde. Aldehydes are frequently involved in self-condensation or polymerization reactions. These reactions are exothermic; they are often catalyzed by acid. Aldehydes are readily oxidized to give carboxylic acids. Flammable and/or toxic gases are generated by the combination of aldehydes with azo, diazo compounds, dithiocarbamates, nitrides, and strong reducing agents. Aldehydes can react with air to give first peroxo acids, and ultimately carboxylic acids. These autoxidation reactions are activated by light, catalyzed by salts of transition metals, and are autocatalytic (catalyzed by the products of the reaction). The addition of stabilizers (antioxidants) to shipments of aldehydes retards autoxidation.
위험도
Flammable, dangerous fire risk. Strong irritant.
건강위험
TOXIC; may be fatal if inhaled, ingested or absorbed through skin. Inhalation or contact with some of these materials will irritate or burn skin and eyes. Fire will produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution.
화재위험
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion and poison hazard indoors, outdoors or in sewers. May polymerize explosively when heated or involved in a fire. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water.
Safety Profile
Poison by ingestion and
sktn contact. Moderately toxic by inhalation.
Severe eye and skin irritant. Mutation data
reported. Dangerously flammable liquid
when exposed to heat, flame, or oxidizers.
Can react vigorously with oxidizing
materials. To fight fire, use CO2, alcohol
foam, foam, dry chemical. When heated to
decomposition it emits acrid smoke and
irritating fumes. See also ALDEHYDES.
Purification Methods
Fractionally distil it under nitrogen through a short Vigreux column (p 11). Store it in sealed ampoules. (Slight polymerisation may occur.) [Beilstein 1 IV 3455.]
메타크롤레인 준비 용품 및 원자재
원자재
준비 용품
3,8-DIMETHYL-1,10-PHENANTHROLINE
3,8-디메틸퀴놀린
FENPROPIMORPH
2-메틸데카날
2,4-다이메틸,1,3-다이옥살론
2-히드록시테트라히드로푸란
2-메탈릴 아세트산
퀴네메라크
아크롤레인 디머, 스테빌라이즈
2,5-DICHLORO-3-METHYLPYRIDINE
3,4-에폭시-2-메틸-1-부텐
2,3-dichloro-2-methylpropionaldehyde
6-메틸-[1,4]디아제판
2-Propen-1-ol, 2-methyl-, 1-formate