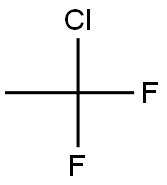
디플루오르모노클로로에탄
|
|
디플루오르모노클로로에탄 속성
- 녹는점
- −131 °C(lit.)
- 끓는 점
- −10 °C(lit.)
- 밀도
- 1.108
- 증기 밀도
- 3.49 (vs air)
- 증기압
- 2196 mm Hg ( 21 °C)
- 굴절률
- 1.3672 (estimate)
- 폭발한계
- 18%
- 안정성
- 안정적인. 가연성이 높습니다. 강한 산화제, 금속과 호환되지 않습니다. 보관에는 황동 조절기, 강철 실린더를 사용하십시오.
- LogP
- 1.330 (est)
- CAS 데이터베이스
- 75-68-3(CAS DataBase Reference)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | F+,N,Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 12-59 | ||
| 안전지침서 | 38-59 | ||
| 유엔번호(UN No.) | UN 2517 2.1 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | KH7650000 | ||
| 위험 참고 사항 | Irritant | ||
| DOT ClassificationII | 2.1 (Flammable gas) | ||
| 위험 등급 | 2.1 | ||
| HS 번호 | 29033990 | ||
| 유해 물질 데이터 | 75-68-3(Hazardous Substances Data) | ||
| 독성 | LC50 inhalation in mouse: 1758gm/m3/2H | ||
| 기존화학 물질 | KE-05597 |
디플루오르모노클로로에탄 C화학적 특성, 용도, 생산
화학적 성질
colourless gas용도
Please view www.aldrich.com/epaods regarding the EPA′s request for application information of Ozone Depleting Substances생산 방법
Chlorodifluoroethane is prepared by the chlorination of difluoroethane in the presence of a suitable catalyst; hydrochloric acid is also formed. The chlorodifluoroethane is purified to remove all traces of water and hydrochloric acid, as well as traces of the starting and intermediate materials.일반 설명
A colorless, odorless gas shipped as a liquid under own vapor pressure. Contact with the unconfined liquid may cause frostbite by evaporative cooling. Easily ignited. Vapors heavier than air. A leak may be either liquid or vapor. May asphyxiate by the displacement of air. Prolonged exposure to fire or intense heat may cause the containers to violently rupture and rocket.공기와 물의 반응
Highly flammable.반응 프로필
1-Chloro-1,1-difluoroethane is chemically inert in many situations, but can react violently with strong reducing agents such as the very active metals and the active metals. Can react with strong oxidizing agents or weaker oxidizing agents under extremes of temperature.위험도
Flammable gas. Explosive limits in air 9.0– 14.8%.건강위험
Vapors may cause dizziness or asphyxiation without warning. Some may be irritating if inhaled at high concentrations. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire may produce irritating and/or toxic gases.화재위험
EXTREMELY FLAMMABLE. Will be easily ignited by heat, sparks or flames. Will form explosive mixtures with air. Vapors from liquefied gas are initially heavier than air and spread along ground. CAUTION: Hydrogen (UN1049), Deuterium (UN1957), Hydrogen, refrigerated liquid (UN1966) and Methane (UN1971) are lighter than air and will rise. Hydrogen and Deuterium fires are difficult to detect since they burn with an invisible flame. Use an alternate method of detection (thermal camera, broom handle, etc.) Vapors may travel to source of ignition and flash back. Cylinders exposed to fire may vent and release flammable gas through pressure relief devices. Containers may explode when heated. Ruptured cylinders may rocket.Pharmaceutical Applications
Chlorodifluoroethane is a hydrochlorofluorocarbon (HCFC) aerosol propellant previously used in topical pharmaceutical formula- tions. However, it is no longer permitted for use in pharmaceutical formulations because of its harmful effects on the environment. It was also generally used in conjunction with difluoroethane to form a propellant blend with a specific gravity of 1. Chlorodifluoroethane was also used in combination with chlorodifluoromethane and hydrocarbon propellants. Chlorodifluoroethane may be used as a vehicle for dispersions and emulsions.Safety Profile
Very ddly toxic by inhalation. Mutation data reported. A very dangerous fire hazard when exposed to heat, flame, or oxidzing materials. To fight fire, stop flow of gas. Can react vigorously with oxidizing materials. When heated to decomposition it emits toxic fumes of Fand Cl-.Safety
Chlorodifluoroethane is no longer permitted for use as an aerosol propellant in topical pharmaceutical formulations. It is generally regarded as an essentially nontoxic and nonirritant material.Deliberate inhalation of excessive quantities of chlorofluorocarbon propellant may result in death, and the following ‘warning’ statements must appear on the label of all aerosols:
WARNING: Avoid inhalation. Keep away from eyes or other mucous membranes.
(Aerosols designed specifically for oral and nasal inhalation need not contain this statement.)
WARNING: Do not inhale directly; deliberate inhalation of contents can cause death.
or
WARNING: Use only as directed; intentional misuse by deliberately concentrating and inhaling the contents can be harmful or fatal.
Additionally, the label should contain the following information:
WARNING: Contents under pressure. Do not puncture or incinerate container. Do not expose to heat or store at room temperature above 120°F (498℃). Keep out of the reach of children.
In the USA, the Environmental Protection Agency (EPA) additionally requires the following information on all aerosols containing chlorofluorocarbons as the propellant:
WARNING: Contains a chlorofluorocarbon that may harm the public health and environment by reducing ozone in the upper atmosphere.