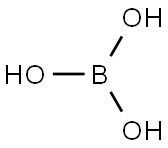
보린산
|
|
보린산 속성
- 녹는점
- >148oC (dec.)
- 저장 조건
- Room Temperature
- 용해도
- DMSO(약간 용해됨), 물(약간 용해됨)
- 물리적 상태
- 액체
- 산도 계수 (pKa)
- 9.2(at 25℃)
- 색상
- 클리어, 다크블루
- InChI
- InChI=1S/BH3O3/c2-1(3)4/h2-4H
- InChIKey
- KGBXLFKZBHKPEV-UHFFFAOYSA-N
- SMILES
- B(O)(O)O
안전
| 기존화학 물질 | KE-03484 | ||
|---|---|---|---|
| 유해화학물질 필터링 | 2019-1-942 | ||
| 중점관리물질 필터링 | 별표1-157 | ||
| 함량 및 규제정보 | 물질구분: 유독물질; 혼합물(제품)함량정보: 붕산[Boric acid; 10043-35-3, 11113-50-1] 및 이를 0.3% 이상 함유한 혼합물 |
보린산 C화학적 특성, 용도, 생산
화학적 성질
Boric acid is a weak acid (pKa = 9.15), existing in aqueous solutions at or below pH 7 as undissociated boric acid. In the solid there is considerable hydrogen bonding between H3BO3 molecules resulting in a layer structure, which accounts for the easy cleavage of the crystals. H3BO3 molecules also exist in dilute solutions but in more concentrated solutions polymeric acids and ions are formed (e.g. H4B2O7; pyroboric acid or tetrahydroxomonoxodiboric(III) acid). The compound is a very weak acid but also acts as a Lewis acid in accepting hydroxide ions:B(OH)3 + H2O→B(OH)4 - + H+
If solid boric acid is heated it loses water and transforms to another acid at 300℃. This is given the formula HBO2 but is in fact a polymer (HBO2)n. It is called metaboric acid or, technically, polydioxoboric(III) acid.
물리적 성질
Boric acid exists in the form of colorless crystals or as a white powder and is soluble in water. It occurs naturally in the condensate from volcanic steam vents (suffioni). Commercially, it is made by treating borate minerals (e.g. kernite, Na2B4O7.4H2O) with sulphuric acid followed by recrystallization.용도
Boric acid is used in the manufacture of glass (borosilicate glass), glazes and enamels, leather, paper, adhesives, and explosives. It is widely used (particularly in the USA) in detergents, and because of the ability of fused boric acid to dissolve other metal oxides it is used as a flux in brazing and welding. Because of its mild antiseptic properties it is used in the pharmaceutical industry and as a food preservative.boric acid is an effective preservative against yeast. It is used in concentrations of 0.01 to 1.0 percent and has fair to good antiseptic properties. It may also be used as a buffer and denaturant. Boric acid is prepared from sulfuric acid and natural borax. It can cause skin rashes and irritation if used in high concentrations. The use of boric acid in cosmetic preparations is no longer very popular.
Indications
Boric Acid is a weakly acidic hydrate of boric oxide with mild antiseptic, antifungal, and antiviral properties. The exact mechanism of action of boric acid is unknown; generally cytotoxic to all cells. It is used in the treatment of yeast infections and cold sores.Boric acid, 600 mg in a gelatin capsule, used intravaginally daily for 14 days, has been reported effective even in resistant Candida infections.
정의
ChEBI: Boric acid is a member of boric acids. It has a role as an astringent. It is a conjugate acid of a dihydrogenborate.제조 방법
Boric acid is prepared by treating borax with nitric acid and nitric acid is chosen over hydrochloric acid because sodium nitrate is more readily soluble than sodium chloride, and sulphuric acid is hard to wash out so not employed.In laboratory boric acid is prepared from sodium borate.
Na2B4O7 + H2SO4 + 5H2O→ Na2SO4 + 4H3BO3
농업용
Boric acid is used as an insecticide for controlling house holding pests like termites, ants, and small insects.Boric acid (H3BO3) is one of the boron-containing nutrients added to fertilizers. It contains around 17% boron. A solution of boric acid and water is used as a foliar spray to overcome boron deficiency.
Pharmaceutical Applications
Boric acid is a long-standing traditional remedy with mainly antifungal and antimicrobial effects. For medicinal uses, it has become known as sal sedativum, which was discovered by Homberg, the Dutch natural philosopher, in 1702. Diluted solutions were and sometimes still are used as antiseptics for the treatment of athletes’ foot and bacterial thrush, and in much diluted solutions as eyewash.잠재적 노출
Boric acid is a fireproofing agent for wood; a preservative, and an antiseptic. It is used in the manufacture of glass, pottery, enamels, glazes, cosmetics, cements, porcelain, borates, leather, carpets, hats, soaps; artificial gems; in tanning leather; printing, dyeing, painting, and photography.운송 방법
UN 3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9—Miscellaneous hazardous material, Technical Name Required.비 호환성
Boric acid decomposes in heat above 100 C, forming boric anhydride and water. Boric acid is hygroscopic; it will absorb moisture from the air. Boric acid aqueous solution is a weak acid; incompatible with strong reducing agents including alkali metals and metal hydrides (may generate explosive hydrogen gas); acetic anhydride, alkali carbonates, and hydroxides. Violent reaction with powdered potassium metal, especially if impacted. Attacks iron in the presence of moisture.폐기물 처리
Boric acids may be recovered from organic process wastes as an alternative to disposal.보린산 준비 용품 및 원자재
원자재
준비 용품
2,3,4,5,6-Pentafluoroaniline
2,5-Dimethylphenylboronic acid
3-(9H-카르바졸-9-일)페닐보론산
6-cyclopropyl-8,9-difluoro-7-methoxy-4-oxo-4,6-dihydro-2H-1l3-[1,3]dioxino[5,6-c]quinoline-2,2-diyl diacetate
Trimethoxyboroxine
Bis(trimethylsilyl)sulfate
TRIPHENYL BORATE
Pinacol
TRIETHANOLAMINE BORATE
6-NITROQUINOLINE
수소
보린산 공급 업체
글로벌( 394)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Zhuanglai Chemical Trading Co.,Ltd | +8613343047651 |
admin@zlchemi.com | China | 2998 | 58 |
| XINGTAI XINGJIU NEW MATERIAL TECHNOLOGY CO., LTD | +8617731987558 |
xingjiu@xingjiubiotech.com | China | 990 | 58 |
| Baoji Guokang Bio-Technology Co., Ltd. | 0917-3909592 13892490616 |
gksales1@gk-bio.com | China | 9316 | 58 |
| Wuhan Ruichi Technology Co., Ltd | +8613545065237 |
admin@whrchem.com | China | 161 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 |
sales@hbmojin.com | China | 12446 | 58 |
| Dorne Chemical Technology co. LTD | +86-86-13583358881 +8618560316533 |
Ethan@dornechem.com | China | 3137 | 58 |
| Firsky International Trade (Wuhan) Co., Ltd | +8615387054039 |
admin@firsky-cn.com | China | 436 | 58 |
| Anhui Yiao New Material Technology Co., Ltd | +86-18033737140 +86-17354101231 |
sales1@hbganmiao.com | China | 253 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 |
deasea125996@gmail.com | China | 2503 | 58 |
| Guangzhou Tengyue Chemical Co., Ltd. | +86-86-18148706580 +8618826483838 |
evan@tyvovo.com | China | 152 | 58 |
보린산 관련 검색:
아세트산 엽산 4-피리딘붕소산 시트르산
Hyaluronic acid
Ascoric Acid
4-tert-Butylphenylboronic acid
4-Carboxyphenylboronic acid
3,4-Difluorophenylboronic acid
3,5-Difluorophenylboronic acid
2-Trifluoromethylphenylboronic acid
3,4,5-Trifluorophenylboronic acid
2,4-Dichlorophenylboronic acid
4-Trifluoromethylphenylboronic acid
4-Cyanophenylboronic acid
BORIC ACID B-10 ENRICHED
BORIC ACID-D3
Boric acid, strontium salt