- Schisandrin C
-
- $0.00 / 20mg
-
2023-02-24
- CAS:61301-33-5
- Min. Order: 5mg
- Purity: ≥98%(HPLC)
- Supply Ability: 10 g
- WUWEIZISU C
-

- $1.00 / 1KG
-
2019-07-06
- CAS: 61301-33-5
- Min. Order: 1g
- Purity: 99%
- Supply Ability: 100KG
|
| | WUWEIZISU C Basic information |
| Product Name: | WUWEIZISU C | | Synonyms: | SCHIZANDRIN C;WUWEIZISU C;,8-dimethyl-;cycloocta(1,2-f:3,4-f’)bis(1,3)benzodioxole6,7,8,9-tetrahydro-1,13-dimethoxy-7;(6R,7S,13aS)-5,6,7,8-Tetrahydro-13,14-dimethoxy-6,7-dimethylcycloocta[1,2-f:3,4-f']bis[1,3]benzodioxole;(S)-(-)-Schisandrin C;Schisandrin C
(S)-(-)-Schisandrin C;13,14-dimethoxy-6,7-dimethyl-5,6,7,8-tetrahydro[1,3]benzodio... | | CAS: | 61301-33-5 | | MF: | C22H24O6 | | MW: | 384.42 | | EINECS: | | | Product Categories: | chemical reagent;pharmaceutical intermediate;phytochemical;reference standards from Chinese medicinal herbs (TCM).;standardized herbal extract;Miscellaneous Natural Products | | Mol File: | 61301-33-5.mol | 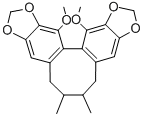 |
| | WUWEIZISU C Chemical Properties |
| Melting point | 122-123℃ | | Boiling point | 549.2±50.0 °C(Predicted) | | density | 1.232 | | solubility | DMSO : 8.33 mg/mL (21.67 mM; Need ultrasonic)H2O : < 0.1 mg/mL (insoluble) | | form | powder | | color | White | | LogP | 5.623 (est) |
| | WUWEIZISU C Usage And Synthesis |
| Description | Schisandrin C is a lignan originally isolated from Schizandrae that has diverse biological activities. It decreases viability of U937 cells in a concentration-dependent manner and induces cell cycle arrest at the G1 phase when used at a concentration of 100 μM. Schisandrin C (5-20 μM) decreases hydrogen peroxide-induced cell death and production of reactive oxygen species (ROS) in C2C12 skeletal muscle cells. It also decreases levels of matrix metalloproteinase-2 (MMP-2), MMP-9, COX-2, VCAM-1, IL-1β, and TNF-α in hydrogen peroxide-stimulated C2C12 cells. Schisandrin C decreases lipoteichoic acid-induced production of nitric oxide (NO), prostaglandin E2 (PGE2; ), TNF-α, IL-1β, and IL-6 in mouse primary microglia. In vivo, schisandrin C (200 mg/kg) decreases serum alanine amino transferase (ALT) and aspartate amino transferase (AST) activity, increases hepatic mitochondrial and total glutathione (GSH) levels, and reduces liver injury in a mouse model of acetaminophen-induced liver injury partially via inhibition of the cytochrome P450 (CYP) isoforms CYP2E1, CYP1A2, and CYP3A11. | | Uses | Schisandrin C potently inhibits proprotein convertase subtillsin-kexin type 9 (PCSK9) mRNA expression. Also, it displays strong protective effects in SH-SY5Y cells against serum and glucose deprivation (SGD) injury. |
| | WUWEIZISU C Preparation Products And Raw materials |
|