|
|
| | Ethyl 3-amino-4,4,4-trifluorocrotonate Basic information |
| Product Name: | Ethyl 3-amino-4,4,4-trifluorocrotonate | | Synonyms: | Ethyl 3-amino-4,4,4-trifluorocrotote;ETHYL 3-AMINO-4,4,4-TRIFLUORO-2-BUTENOATE;ETHYL 3-AMINO-4,4,4-TRIFLUOROBUT-2-ENOATE;ETHYL 3-AMINO-4,4,4-TRIFLUOROCROTONATE;2-BUTENOIC ACID, 3-AMINO-4,4,4-TRIFLUORO, ETHYL ESTER;3-AMINO-4,4,4-TRIFLUOROCROTONIC ACID ETHYL ESTER;3-AMINO-4,4,4-TRIFLUOROCROTONIC ETHYL ESTER;Ethyl 3-Amino-4,4,4-Trifluorocrotonate Ethyl 3-Amino-4,4,4-Trifluoro-2-Butenoate | | CAS: | 372-29-2 | | MF: | C6H8F3NO2 | | MW: | 183.13 | | EINECS: | 609-360-9 | | Product Categories: | Fluorinated ketene derivatives;Ketene derivatives | | Mol File: | 372-29-2.mol | 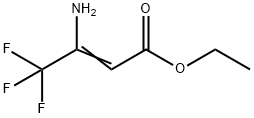 |
| | Ethyl 3-amino-4,4,4-trifluorocrotonate Chemical Properties |
| Melting point | 26°C | | Boiling point | 83 °C15 mm Hg(lit.) | | density | 1.245 g/mL at 25 °C(lit.) | | refractive index | n20/D 1.424(lit.) | | Fp | 149 °F | | storage temp. | 2-8°C(protect from light) | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Colourless to Pale Yellow Low-Melting to Semi-Solid | | pka | 0.76±0.70(Predicted) | | color | White or Colorless to Light yellow | | FreezingPoint | 22.0 to 26.0 ℃ | | Sensitive | Air Sensitive | | BRN | 4397839 | | InChI | 1S/C6H8F3NO2/c1-2-12-5(11)3-4(10)6(7,8)9/h3H,2,10H2,1H3 | | InChIKey | NXVKRKUGIINGHD-ONEGZZNKSA-N | | SMILES | CCOC(=O)C=C(N)C(F)(F)F | | CAS DataBase Reference | 372-29-2(CAS DataBase Reference) |
| Hazard Codes | Xn,T,Xi | | Risk Statements | 20/21/22-36/37 | | Safety Statements | 26-36-36/37 | | RIDADR | 3259 | | WGK Germany | 3 | | Hazard Note | Toxic/Irritant | | TSCA | TSCA listed | | HazardClass | IRRITANT | | HazardClass | 8 | | HS Code | 29224999 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Acute Tox. 4 Dermal
Acute Tox. 4 Oral
Eye Irrit. 2
STOT SE 3 |
| | Ethyl 3-amino-4,4,4-trifluorocrotonate Usage And Synthesis |
| Chemical Properties | Colourless liquid or white solid | | Uses | Ethyl 3-amino-4,4,4-trifluorocrotonate is used as building block in chemical synthesis. Attributes Development Product Contact | | Synthesis | A slurry of sodium enolides was prepared from ethyl trifluoroacetate (88.1 g, 1.0 mol) and ethyl acetate (71.05 g, 0.50 mol) by mixing with solid sodium ethoxide (34.05 g, 0.5 mol). The mixture was evaporated and concentrated with reference to the operation of Example 1. Subsequently, 250 ml of cyclohexane was added to the reaction system, followed by ammonium acetate (77.1 g, 1.0 mol) and anhydrous acetic acid (39.0 g, 0.65 mol). The resulting suspension was heated to boiling, water was separated by distillation and cyclohexane was refluxed into the reaction mixture. The reaction was terminated after 5 hours. The organic phase was separated by adding 300 mL of water. Cyclohexane was removed from the organic phase by distillation and the product was subsequently fractionated under vacuum. 57.0 g of ethyl 3-amino-4,4,4-trifluorobut-2-enoate was obtained with 97.4% purity and 62% yield. The product was analyzed by EI mass spectrometry ([M+] = 183 AMU), 1H-NMR (7.6 ppm, NH; 4.86 ppm, 1H, CH; 4.08 ppm, Quadruple peak, 2H, ethyl; 1.18 ppm, Triple peak, 3H, ethyl) and 13C-NMR (168 ppm, COOEt; 147 ppm, Quadruple peak, C-NH2. 120 ppm, broad quadruple peak, CF3; 82 ppm, quadruple peak, CH; 59 ppm, ethyl; 14 ppm, ethyl) were characterized. | | References | [1] Patent: WO2004/16579, 2004, A1. Location in patent: Page 9; 10 |
| | Ethyl 3-amino-4,4,4-trifluorocrotonate Preparation Products And Raw materials |
|