Talniflumate manufacturers
|
| | Talniflumate Basic information |
| Product Name: | Talniflumate | | Synonyms: | 2-(alpha,alpha,alpha-trifluoro-m-toluidino)-nicotinicaciphthalidylester;3-ftalidilicodelacido2-((3-(trifluormetil)fenil)amino)-3-piridincarboxilic;3-phthalidylesterof2-((3-(trifluoromethyl)phenyl)amino)-3-pyridinecarboxyl;Talniflumate, >=99%;2-(alpha,alpha,alpha-trifluoro-m-toluidino)nicotinicacidphthalidylester;ba7602-06;phthalidyl2-(3-trifluoromethylanilino)nicotinate;phthalidyl2-(alpha,alpha,alpha-trifluoro-m-toluidino)nicotinate | | CAS: | 66898-62-2 | | MF: | C21H13F3N2O4 | | MW: | 414.33 | | EINECS: | 1533716-785-6 | | Product Categories: | | | Mol File: | 66898-62-2.mol | 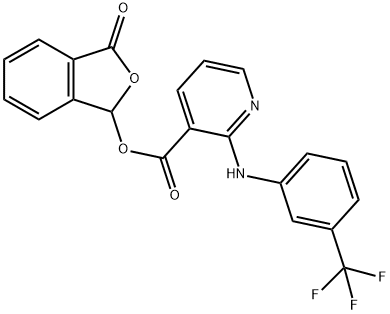 |
| | Talniflumate Chemical Properties |
| Melting point | 165-167°C | | Boiling point | 534.6±50.0 °C(Predicted) | | density | 1.49±0.1 g/cm3(Predicted) | | storage temp. | Store at +4°C | | solubility | Chlorform (Slightly), DMSO (Slightly), Methanol (Slightly) | | pka | 2.72±0.10(Predicted) | | form | Solid | | color | Off-White to Pale Yellow | | λmax | 287nm(CHCl3)(lit.) | | Merck | 14,9044 | | Stability: | Acid Sensitive, Moisture Sensitive |
| RTECS | QT2999200 | | HS Code | 2933.39.9200 | | Toxicity | LD50 orally in rats: 12000 mg/kg (Los) |
| | Talniflumate Usage And Synthesis |
| Chemical Properties | White to pale yellow crystalline powder | | Originator | Somalgen,Bago,Argentina,1972 | | Uses | Anti-inflammatory; analgesic. | | Uses | Talniflumate is a nonaspirin and nonsteroidal anti-inflammatory drug. | | Definition | ChEBI: 2-[3-(trifluoromethyl)anilino]-3-pyridinecarboxylic acid (3-oxo-1H-isobenzofuran-1-yl) ester is a member of benzofurans. | | Manufacturing Process | 49 ml of triethylamine were added to a suspension of 2-(3'-
trifluoromethylanilino)nicotinic acid (70.6 g in 250 ml of dimethylformamide).
After stirring for 30 minutes 53.3 g of 3-bromophthalide were added. The
reaction mixture was maintained at 25°C to 30°C during 4 hours. Ethyl
acetate (750 ml) was poured into the reaction mixture. This solution was
filtered and extracted with water (4 x 250 ml), discarding the water layer.
The organic layer was dried with anhydrous magnesium sulfate and then
filtered. The solution was concentrated under vacuum at 30°C to 35°C until
reduced to half of its original volume and then cooled to 5°C to allow the
crystallization of the compound. Thus, the cake was filtered, washed with cool
ethyl acetate, and dried under vacuum. Yield: 74% (76.7 g) of phthalidyl
ester of 2-(3'-trifluoromethylanilino)-pyridin-3-carboxylic acid, melting point:
165°C to 167°C. | | Brand name | Somalgen (Laboratorio Bago, S.A., Argentina). | | Therapeutic Function | Antiinflammatory, Analgesic | | Biological Activity | Calcium-activated chloride channel (hCLCA1/mCLCA3) blocker; reduces mucin synthesis and release in cell culture and animal models. Possesses anti-inflammatory actions via inhibition of cyclooxygenases and inhibits Cl - /HCO 3 - exchanger activity. Increases survival in a cystic fibrosis mouse model of distal intestinal obstructive syndrome. |
| | Talniflumate Preparation Products And Raw materials |
|