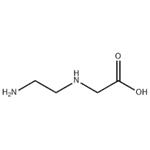
- N-(2-AMINOETHYL)GLYCINE
-
- $34.00 / 1kg
-
2025-09-25
- CAS:24123-14-6
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | N-(2-AMINOETHYL)GLYCINE Basic information |
| | N-(2-AMINOETHYL)GLYCINE Chemical Properties |
| Melting point | 140-143°C | | Boiling point | 290℃ | | density | 1.161 | | Fp | 129℃ | | storage temp. | Keep in dark place,Sealed in dry,2-8°C | | solubility | Methanol (Slightly, Sonicated), Water (Slightly) | | pka | 2.21±0.10(Predicted) | | form | Solid | | color | White to Off-White | | Water Solubility | very faint turbidity | | Stability: | Incompatible with strong oxidizing agents. | | InChI | InChI=1S/C4H10N2O2/c5-1-2-6-3-4(7)8/h6H,1-3,5H2,(H,7,8) | | InChIKey | PIINGYXNCHTJTF-UHFFFAOYSA-N | | SMILES | C(O)(=O)CNCCN |
| Hazard Codes | Xn,Xi | | Risk Statements | 20/21/22-36/37/38-36 | | Safety Statements | 24/25-36-26 | | WGK Germany | WGK 3 | | HS Code | 2922499990 | | Storage Class | 11 - Combustible Solids | | Hazard Classifications | Eye Irrit. 2 |
| | N-(2-AMINOETHYL)GLYCINE Usage And Synthesis |
| Chemical Properties | White Solid | | Uses | 2-((2-Aminoethyl)amino)acetic acid is a Glycine.html" class="link-product" target="_blank">Glycine (HY-Y0966) derivative[1]. | | Synthesis | The general procedure for the synthesis of 2-((2-aminoethyl)amino)acetic acid from ethylenediamine and chloroacetic acid is as follows:
Example 8: Preparation of [(2-aminoethyl)amino]acetic acid (3) in DMSO
1. Preparation of chloroacetic acid solution: chloroacetic acid (500.0 g, 5.29 mol) was dissolved in DMSO (515.6 g).
2. Reaction setup: ethylenediamine (3537mL, 52.9mol) was added to a 5L flask equipped with a nitrogen inlet, mechanical stirrer, thermocouple and peristaltic metering pump. Begin cooling with ice water.
3. Dropwise addition of chloroacetic acid solution: chloroacetic acid solution in DMSO was slowly added to ethylenediamine via a metering pump. The reaction conditions are shown in Table 1.
Table 1: Reaction conditions during addition of chloroacetic acid solution to ethylenediamine
| No. | Time | Temperature (°C) | Pressure (Torr) |
|------|--------|----------|-----------|
| 1 | 10:51 | 17 | - |
| 2 | 11:28 | 18 | - |
| 3 | 12:18 | 16 | 720 |
| 4 | 13:38 | 11 | 625 | 4 | 13:38 | 11 | 625
| 5 | 14:35 | 12 | 550 | 4 | 13:38 | 11 | 625 | 4
6 | 15:17 | 10 | 490 | 7 | 16:08 | 16:08
| 7 | 16:08 | 8 | 420 | 8 | 8:59 | 8 | 8:59 | 8 | 8:59 | 8 | 8:59
| 8 | 8:59* | 19 | 0 |
*Time represents the next day.
4. Concentration of reaction mixture: The reaction mixture was concentrated by rotary evaporation (50-60 °C, 14-21 Torr) to remove ethylenediamine. The total amount of distillate collected was 2400mL.
5. Toluene treatment: Toluene (900 mL) was added to the residue and the mixture was further concentrated by rotary evaporation (60 °C, 30 Torr).
6. Transfer and cooling: The residue was transferred to a 5L flask using DMSO (3kg). The internal temperature was 28°C and the mixture was cloudy. Cooling (ice/water) was started.
7. Overnight stirring: the reaction mixture was stirred overnight.
8. filtration and washing: the resulting suspension was filtered through filter paper. The flask and solid were washed with DMSO (2 x 500mL). The solid was subsequently washed with isopropanol (2 x 500mL) and tert-butyl methyl ether (2 x 500mL).
9. Drying: The solid was dried at ambient temperature under high vacuum for 18 h. 10.
10. Product collection: 2-((2-aminoethyl)amino)acetic acid (3) was obtained as a white solid (480.4 g, 4.07 mol) in 77% yield. | | References | [1] Luckose F, et al. Effects of amino acid derivatives on physical, mental, and physiological activities. Crit Rev Food Sci Nutr. 2015;55(13):1793-1144. DOI:10.1080/10408398.2012.708368 |
| | N-(2-AMINOETHYL)GLYCINE Preparation Products And Raw materials |
|